
• Some naturally occuring acids
Vinegar – Acetic Acid
Orange – Citric Acid
Lemon – Citric Acid
Tamarind – Tartaric Acid
Tomato – Oxalic Acid
Sour milk (Curd) – Lactic Acid
Ant and Nettle sting – Methanoic Acid
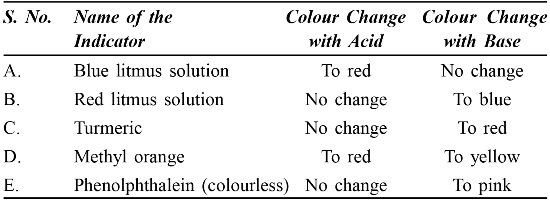
• Acid – Base Indicators – Indicate the presence of an acid or base in a solution.
• Litmus solution – It is a natural indicator. It is a purple dye extracted from Lichens. Other examples are Red Cabbage and coloured petals of Petunia and turmeric.
• Olfactory indicators – Show odour changes in acidic or basic media.
eg. onion and clove.
• Acid – Base Indicators
• Dilute Acid : Contains only a small amounts of acid and a large amount of water.
• Concentrated Acid : A concentrated acid contains a large amount of acid and a small amount of water.
• Chemical Properties of Acids and Bases
(i) Reaction with metal
Acid + Metal → Salt + Hydrogen
2HCl + Zn → ZnCl2 + H2
2HNO3 + Zn → Zn (NO3)2 + H2
H2SO4 + Zn → ZnSO4 + H2
2CH3COOH + Zn → (CH3COO)2 Zn + H2
• Pop test : When a burning candle is brought near a test tube containing hydrogen gas it burns with a ‘Pop’ sound. This test is conducted for examining the presence of hydrogen gas.
• Base + Metal → Salt + Hydrogen
NaOH + Zn → Na2ZnO2 + H2
Sodium Zincate
Note – Such reactions are not possible with all the metals.
Latest Govt Job & Exam Updates: