PART II : CHEMISTRY
SECTION I : (Only One option correct Type)
This section contains 10 multiple choice questions. Each question has four choices (A), (B), (C) and (D) out of which ONLY ONE is correct
1. Consider the following complex ions, P, Q and R.
P = [FeF6]−3, Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+
The correct order of the complex ions, according to their spin−only magnetic moment values (in B.M.) is
(A) R< Q < P
(B) Q < R < P
(C) R < P < Q
(D) Q < P < R
2. The arrangement of X− ions around A+ ion in solid AX is given in the figure (not drawn to scale). If the radius of X− is 250 pm, the radius of A+ is

(A) 104 pm
(B) 125 pm
(C) 183 pm
(D) 57 pm
3. Sulfide ores are common for the metals
(A) Ag, Cu and Pb
(B) Ag, Cu and Sn
(C) Ag, Mg and Pb
(D) Al, Cu and Pb
4. The standard enthalpies of formation of CO2(g), H2O (ℓ) and glucose (s) at 25℃ are −400 kJ/mol, −300 kJ/mol and −1300 kJ/mol, respectively. The standard enthalpy of combustion per gram of glucose at 25℃ is
(A) +2900 kJ
(B) −2900 kJ
(C) −16.11 kJ
(D) +16.11 kJ
5. Upon treatment with ammonical H2S, the metal ion that precipitates as a sulfide is
(A) Fe(III)
(B) Al(III)
(C) Mg(II)
(D) Zn(II)
6. Methylene blue, from its aqueous solution is adsorbed on activated charcoal at 25° C. for this process, the correct statement is
(A) The adsorption requires activation at 25° C.
(B) The adsorption is accompanied by a decrease in enthalpy.
(C) The adsorption increases with increase of temperature.
(D) The adsorption is irreversible.
7. KI in acetone, undergoes SN2 reaction with each of P, Q, R and S. The rates of the reaction vary as
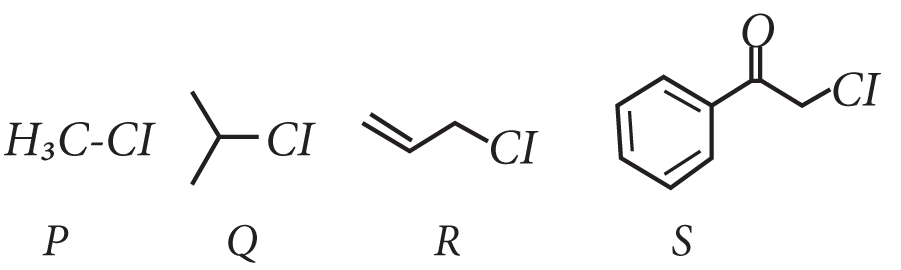
(A) P > Q > R > S
(B) S > P > R > Q
(C) P > R > Q > S
(D) R > P > S > Q
8. In the reaction,
P + Q → R + S
the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. The concentration of Q varies with reaction time as shown in the figure. The overall order of the reaction is
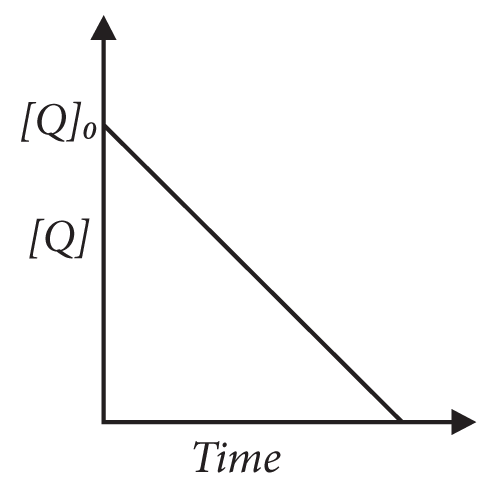
(A) 2
(B) 3
(C) 0
(D) 1
9. Concentrated nitric acid, upon long standing, turns yellow − brown due to the formation of
(A) NO
(B) NO2
(C) N2O
(D) N2O4
10. The compound that does NOT liberate CO2, on treatment with aqueous sodium bicarbonate solution, is
(A) Benzoic acid
(B) Benzenesulphonic acid
(C) Salicylic acid
(D) Carbolic acid (Phenol)
SECTION II : (One or more options correct Type)
This section contains 5 multiple choice questions. Each question has four choices (A), (B), (C) and (D) out of which ONE or MORE are correct.
11. The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1M), at 25°C. the Ka of HA is
(A) 1 × 10−4
(B) 1 × 10−5
(C) 1 × 10−6
(D) 1 × 10−3
12. The hyperconjugative stabilities of tert − butyl cation and 2 − butene, respectively, are due to
(A) σ → p (empty) and σ → π* electron delocalisations.
(B) σ → σ* and σ →π electron delocalisations.
(C) σ → p (filled) and σ → π electron delocalisations.
(D) p(filled) → σ* and σ → π* electron delocalisations.
13. The pair(s) of coordination complexes / ions exhibiting the same kind of isomerism is (are)
(A) [Cr(NH3)5Cl]Cl2 and [Cr(NH3)4Cl2]Cl
(B) [Co(NH3)4Cl2]+ and [Pt(NH3)2 (H2O)Cl]+
(C) [CoBr2Cl2]2− and [PtBr2Cl2]2−
(D) [Pt(NH3)3(NO3)]Cl and [Pt(NH3)3Cl]Br
14. Among P, Q, R and S, the aromatic compound (s) is/are

(A) P
(B) Q
(C) R
(D) S
15. Benzene and naphthalene from an ideal solution at room temperature. For this process, the true statement(s) is(are)
(A) ΔG is positive
(B) ΔSsystem is positive
(C) ΔSsurroundings = 0
(D) ΔH = 0
SECTION III : (Integer value correct Type)
This section contains 5 questions. The answer to each question is a single digit integer, ranging from 0 to 9 (both inclusive).
16. The atomic masses of He and Ne are 4 and 20 a.m.u, respectively. The value of the de Broglie wavelength of He gas at −73°C is “M” times that of the de Broglie wavelength of Ne at 727°C. M is
17. EDTA4− is ethylenediaminetetraacetate ion. The total number of N − Co − O bond angles in [Co(EDTA)]1− complex ion is
Answer: (8)
18. The total number of carboxylic acid groups in the product P is
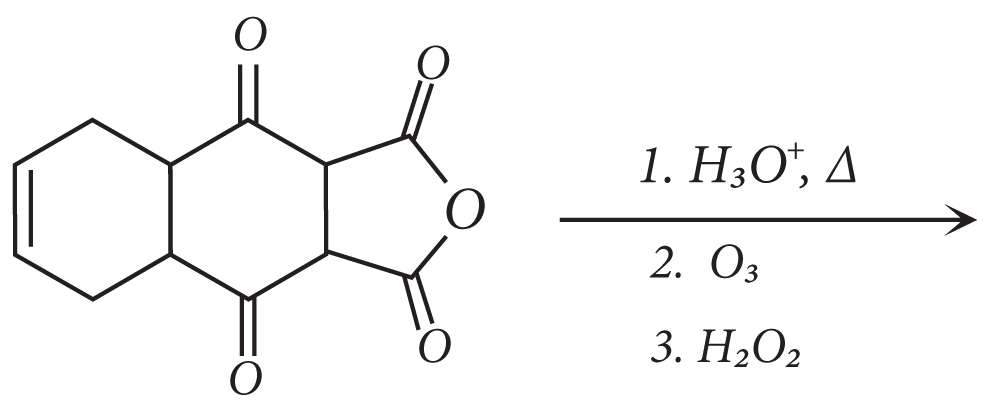
19. A tetrapeptide has − COOH group on alanine. This produces glycine (Gly), valine (Val), phenyl alanine (Phe) and alanine(Ala), on complete hydrolysis. For this tetrapeptide, the number of possible sequences (primary structures) with −NH2 group attached to a chiral center is
20. The total number of lone − pairs of electrons in melamine is
Latest Govt Job & Exam Updates: