Chemistry
SECTION – 1 : (Maximum Marks : 32)
This section contains EIGHT questions
The answer to each question is a SINGLE DIGIT INTEGER ranging from 0 to 9, both inclusive
1. In the complex acetylbromidodicarbonylb is(triethylphosphine)iron(II), the number of Fe–C bond(s) is
2. Among the complex ions, [Co(NH2 – CH2 – CH2 – NH2)2Cl2]+, [CrCl2(C2O4)2]3−, [Fe(H2O)4(OH)2]+, [Fe(NH3)2(CN)4]−,
[Co(NH2 – CH2 – CH2 – NH2)2(NH3)Cl]2+ and [Co(NH3)4(H2O)Cl]2+, the number of complex ion(s) that show(s) cis-trans isomerism is
3. Three moles of B2H6 are completely reacted with methanol. The number of moles of boron containing product formed is
4. The molar conductivity of a solution of a weak acid HX (0.01 M) is 10 times smaller than the molar conductivity of a solution of a weak acid HY (0.10 M). If ![]() the difference in their pKa values, pKa(HX) – pKa(HY), is
the difference in their pKa values, pKa(HX) – pKa(HY), is
(consider degree of ionization of both acids to be <<1)
5. A closed vessel with rigid walls contains 1 mol of ![]() and 1 mol of air at 298 K. Considering complete decay of
and 1 mol of air at 298 K. Considering complete decay of ![]() the ratio final pressure to the initial pressure of the system at 298 K is
the ratio final pressure to the initial pressure of the system at 298 K is
6. In dilute aqueous H2SO4, the complex diaquodioxalatoferrate (II) is oxidized by ![]() . For this reaction, the ratio of the rate of change of [H+] to the rate of change
. For this reaction, the ratio of the rate of change of [H+] to the rate of change ![]() is
is
7. The number of hydroxyl group(s) in Q is
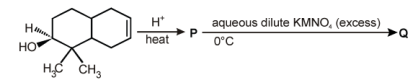
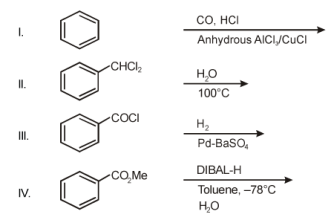
8. Among the following the number of reaction(s) that produce(s) benzaldehyde is
SECTION – 2 : (Maximum Marks : 32)
• This section contains EIGHT questions
• Each question has FOUR options (A), (B), (C) and (D). ONE OR MORE THAN ONE of these four option(s) is(are) correct
9. Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectively, are
(A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
10. When O2 is adsorbed on a metallic surface, electron transfer occurs from the metal to O2. The TRUE statement(s) regarding this adsorption is(are)
(A) ![]() is physisorbed
is physisorbed
(B) heat is released
(C) occupancy of ![]() is increased
is increased
(D) bond length of ![]() is increased
is increased
11. One mole of a monoatomic real gas satisfies the equation p(V – b) = RT where b is a constant. The relationship of interatomic potential V(r) and interatomic distance r for the gas is given by
(A) 
(B) 
(C) 
(D) 
12. In the following reactions, the product S is

(A) 
(B) 
(C) 
(D) 
13. The major product U in the following reactions is

(A) 
(B) 
(C) 
(D) 
14. In the following reactions, the major product W is

(A) 
(B) 
(C) 
(D) 
15. The correct statement(s) regarding, (i) HClO, (ii) HClO2, (iii) HClO3 and (iv) HClO4 is(are)
(A) The number of Cl=O bonds in (ii) and (iii) together is two
(B) The number of lone pairs of electrons on Cl in (ii) and (iii) together is three
(C) The hybridization of Cl in (iv) is sp3
(D) Amongst (i) to (iv), the strongest acid is (i)
16. The pair(s) of ions where BOTH the ions are precipitated upon passing H2S gas in presence of dilute HCl, is(are)
(A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
SECTION – 3 : (Maximum Marks : 16)
• This section contains TWO paragraphs
• Based on each paragraph, there will be TWO questions.
• Each equation has FOUR options (A), (B), (C) and (D). ONE OR MORE THAN ONE of these four option(s) is(are) correct
PARAGRAPH-1
(17 & 18)
In the following reactions :

17. Compound X is
(A) 
(B) 
(C) 
(D) 
18. The major compound Y is
(A) 
(B) 
(C) 
(D) 
PARAGRAPH-2
(19 & 20)
When 100 mL of 1.0 M HCl was mixed with 100 mL of 1.0 M NaOH in an insulated beaker at constant pressure, a temperature increase of 5.7 ºC was measured for the beaker and its contents (Expt. 1). Because the enthalpy of neutralization of a strong acid with a strong base is a constant (–57.0 kJ mol−1), this experiment could be used to measure the calorimeter constant. In a second experiment (Expt. 2), 100 mL of 2.0 M acetic acid (Ka = 2.0 × 10–5) was mixed with 100 mL of 1.0 M NaOH (under identical conditions to Expt. 1) where a temperature rise of 5.6 ºC was measured.
19. Enthalpy of dissociation (in kJ mol–1) of acetic acid obtained from the Expt. 2 is
(A) 1.0
(B) 10.0
(C) 24.5
(D) 51.4
20. The pH of the solution after Expt. 2 is
(A) 2.8
(B) 4.7
(C) 5.0
(D) 7.0
Latest Govt Job & Exam Updates: