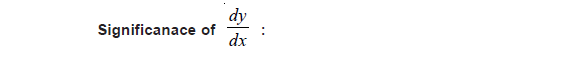CHEMISTRY
Some Basic Concepts of Chemistry
General Introduction :
Importance and scope of chemistry. Historical approach to particulate nature of matter, laws of chemical combination, Dalton’s atomic theory : theory : concept of elements, atoms and molecules. Atomic and molecular masses. Mole concept and molar mass : percentage composition, empirical and molecular formula; chemical reactions, stoichiometry and calculations based on stoichiometry.
Structure of Atom
Discovery of electron, proton and neutron, atomic number, isotopes and isobars. Thomson’s model and its limitations, Rutherford’s model and its limitations. Borh’s model and its limitations, concept of shells and subshells, dual nature of matter and light, De Broglies relationship, Heisenberg uncertainty principle, concept of orbitals, quantum numbers, shapes of s, p, and d orbitals, rules for filling electrons in orbitals-Aufbau principle, Pauli exclusion principle and Hund’s rule, electronic configuration of atoms, stability of half filled and completely filled orbitals.
Classification of Elements and Periodicity in Properties :
Significance and classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements-atomic radii, ionic radii, inert gas radii. Ionization enthalpy, electron gain enthalpy, electro Magnetic Effects of current and Magnetism : Concept of magnetic field, Oersted’s experiment. Biot – Savart law and its application to current carrying circular loop. Ampere’s law and its applications to infinitely long straight wire, straight and toroidal solenoids. Force on a moving charge in uniform magnetic and electric fields. Cyclotron. Force on a current-carrying conductor in a uniform magnetic field. Force between two parallel current-carrying conductors-definition of ampere. Torque experienced by a current loog in uniform magnetic field; moving coil galvanometer-its current sensitivity and conversion to ammeter and voltmeter. Current loog as a magnetic dipole and its magnetic dipole moment. Mangetic dipole moment of a revolving electron. Magnetic field intensity due to magnetic dipole (bar magnet) along its axis and perpendicular to its axis. Torque on a magnetic dipole (bar magnet) in a uniform magnetic field; bar magnet as an equivalent solenoid, magnetic field lines; Earth’s magnetic field and magnetic elements. Para-dia-and ferromagnetic substances, with examples. Electromagnets and factors affecting their stengths. Permanent magnets.
negativity, valence.
Chemical Bonding and Molecular Structure :
Valence electrons, ionic bond, covalent bond: bond parameters. Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR theory, concept of hybridization, involving s, p orbitals and shapes of some simple molecules, molecular orbital; theory of homo nuclear diatomic molecules (qualitative idea only), hydrogen bond.
States of Matter : gases and liquids :
Three states of matter. Intermolecular interactions, type of bonding, melting and boiling points. Role of gas laws in
elucidating the concept of the molecule, Boyle’s law, Charles law, Gay Lussac’s law, Avogadro’s law, ldeal behaviour, empirical derivation of gas equation, Avogadro’s number. Ideal gas equation. Derivation from ideal behaviour, liquefaction of gases, critical temperature. Liquid State-Vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations.)
Thermodynamics :
Concepts of System, types of systems, surroundings. Work, heat, energy, extensive and intensive properties, state functions. First law of thermodynamics – internal energy and enthalpy, heat capacity and specific heat, measurement of ÎU and ÎH, Hess’s law of constant heat summation, enthalpy of: bond dissociation, combustion, formation, atomization, sublimation. Phase transition, ionization, and dilution. Introduction of entropy as a state function, free energy change for spontaneous and non-spontaneous process, equilibrium.
Equilibrium :
Equilibrium in physical and chemical processes, dynamic nature e of equilibrium, law of mass action, equilibrium constant, factors affecting equilibrium – Le Chatelier’s principle; ionic equilibrium – ionization of acids and bases, strong and weak electrolytes, degree of ionization, concept of pH. Hydrolysis of salts (elementary idea). Buffer solutions. Solubility product, common ion effect (with illustrative examples).
Redox Reactions :
Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, applications of redox reactions.
Hydrogen :
Position of hydrogen in periodic table, occurrence, isotopes, preparation, properties and uses of hydrogen; hydrides – ionic, covalent and interstitial, physical and chemical properties of water, heavy water, hydrogen peroxide-preparation, reactions and structure; hydrogen as a fuel .
S-Block Elements (Alkali and Alkaline earth metals)
Group 1 and Group 2 elements :General introduction, electronic configuration, occurrence, anomalous properties of the first element of each group, diagonal relationship, trends in the variation of properties (such as ionization enthalpy, atomic and ionic radii), trends in chemical reactivity with oxygen, water, hydrogen and halogens; uses.
Preparation and properties of some important compounds :
Sodium carbonate, sodium chloride, sodium hydroxide and sodium hydrogen carbonate, biological importance of sodium and potassium. CaO, CaCO3 and industrial use of lime and limestone, biological importance of Mg and Ca
Some P-Block Elements :
General introduction to p-Block Elements Group 13 elements : General introduction, electronic configuration, occurrence, Variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of first element of the group; Boron-physical and chemical properties, some important compounds: borax, boric acids, boron hydrides. Aluminium : uses, reactions with acids and alkalies. Group 14 elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous behaviour of first element, Carbon – catenation, allotropic forms, physical and chemical properties; uses of some important compounds: oxides. Important compounds of silicon and a few uses: silicon tetrachloride, silicones, silicates and zeolites. Organic Chemistry – Some Basic Principles and Techniques General introduction method, qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds Electronic displacements in a covalent bond: free radicals, carbocations, carbanions; heterophiles and nucleophiles, types of organic reactions.
Hydrocarbons :
Classification of hydrocarbons Alkanes-Nomenclature, isomerism, conformations (ethane only), physical roperties, chemical reactions including free radical mechanism or halogenation, combustion and pyrolysis. Alkenes-Nomenclature, structure of double bond (ethene) geometrical isomerism, physical properties, methods f preparation, chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikov’s a dition and peroxide effect), ozonolysis, oxidation mechanism of electrophilic addition. Alkynes-Nomenclature, structure of triple bond (ethyne), physical properties, Methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of -hydrogen, halogens, hydrogen halides and water. Aromatic hydrocarbons : Introduction, IUPAC nomenclature; Benzene: resonance aromaticity; chemical
properties : mechanism of electrophilic substitution. – nitration sulphonation, halogenation, Friedel Craft’s alkylation and acylation: directive influence of functional group in mono-substituted benzene; carcinogenicity and
t xicity.
Environmental Chemistry :
Environmental pollution – air, water and soil pollution, chemical reactions in tmosphere, smog, major atmospheric pollutants; acid rain, ozone and its reactions effect of depletion of ozone layer, greenhouse effect and global warming – pollution due to industrial wastes; green chemistry as an alternative ool for reducing pollution strategy for control of environmental pollution.
Solid State :
Classification of solids based on different binding forces: molecular, ionic, covalent and metallic solids, amprophous and crystalline solids (elementary idea), unit cell in two
dimensional a nd three dimensional lattices, calculation of density of unit cell, packing in solids, voids, number of
atoms per unit cell in a cubic unit cell, point defects, electrical and magnetic properties.
Solutions :
Types of olutions, expression of concentration of solutions of solids in liquids, solubility of gases in liquids, solid solutions, olligative properties -relative lowering of vapour pressure, elevation of Boiling Point, depression of freezing point, osmotic pressure, determination of molecular masses using colligative properties, abnormal molecular mass.
Electrochemistry :
Redox reactions, conductance in electrolytic solutions, specific and molar conductivity variations of conductivity with concentration, Kohlrausch’s Law, electrolysis and laws of electrolysis (elementary idea), dry cell-electrolytic cells and Galvanic cells; lead accumulator, EMF of a cell, standard electrode potential, Nernst equation and its application to chemical cells, fuel cells; corrosion.
Chemical Kinetics :
Rate of a reaction (average and instantaneous), factors affecting rates of reaction; concentration, temperature, catalyst, order and molecularity of a reaction, rate law and specific rate constant, integrated rate quations and half life (only for zero and first order reactions); concept of collision theory (elementay idea, no mathematical treatment)
Surface Chemistry :
Adsorption – physisorption and chemisorption; factors affecting adsorption of gases on solids; catalysis: homogenous and heterogeneous, activity and selectivily: enzyme catalysis; colloidal state: Distinction between true solutions, colloids and suspensions; lyophilic, lyophobic, multimolecular and macromolecular clloids; properties of colloids; Tyndall effect, Brownian movement, electrophoresis, coagulation; emulsion – typesof emulsions.
Generla Principles and Processes of lsolation of Elements :
Principles and methods of extraction – concentration, oxidation, reduction electrolytic method and refining; occurrence and principles of extraction of aluminium, copper, zinc and lron….
P-Block Elements :
Group 15 elements : General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; nitrogen – perparation, properties and uses; compounds of nitrogen preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only) Phosphorous-allotropic forms; compounds of phosphorous: preparation and properties of phosphine, halides (PCI3, PCI5) and oxoacids (elementary idea only)
Group 16 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemicla properties, dioxygen: preparation, properties and uses; simple oxides; Ozone. Sulphurallotropic forms; compounds of sulphur: preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Group 17 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric aoid, interhalogen compounds, oxoacids of halogens (structures only)
Group 18 elements : General introduction, electronic configuration. Occurrence, trends in physical and chemicla properties, uses.
D and F Block Elemets :
General introduction, electronic configuration, occurrence and characteristics of transition metals, general trends in properties of the first row transition metals – metallic character, ionization enthalpy, oxidation states, ionic radii, colour catalytic property, magnetic properties, interstitial compounds, alloy formation. Preparation and properties of K2Cr2O7 and KMnO4
Lanthanoids – electronic configuration, oxidation states, chemical reactivity and lanthanoid contraction.
Actinoids – Electronic configuration, oxidation states,
Coordination Compounds :
Coordination compounds – Introduction, ligands, coordination number colour, magnetic properties and shapes. IUPAC nomenclature of mononuclear coordination compounds. bonding; isomerism, importance of coordination compounds (in qualitative analysis, extraction of metals and biological systems).
Haloalkanes and Haloarenes.
Haloalkanes:
Nomenclature, nature of C-X bond, physical and chemical properties mechanism of substitution reactions.
Haloarenes :
Nature of C-X bond, substitution reactions (direvtive influence of halogen for monosubstituted compounds only) Uses and environmental effects of – dichloromethane, trichloromethane, tetrachloromethane, iodoform, freons, DDT,
Alcohols, Phenols and Ethers :
Ahenols: Nomenclature, methods of preparation, physical and chemical properties (of primary alcohols only); identification of primary, secondary and tertiary alcohols; mechanism of dehydration, uses of methanol and ethanol.
Phenols : Nomenclature, methods of preparation, physical and chemical properties, acidic nature of phenol,
electrophillic substitution reactions, uses of phenols.
Ethers: Nomenclature, methods of preparation, physical and chemical properties, uses.
Aldehydes, Ketones and Carboxylic Acids
Aldehydes and Ketones : Nomenclature, nature of carbonyl group methods of preparation, physical and chemical properties mechanism of nucleophilic addition, reactivity of alpha hydrogen in aldehydes; uses.
Caboxylic Acids : Nomenclature, acidic nature, methods. of preparation, physical and chemical porperties; uses.
Organic compounds containing Nitrogen
Amines: Namenclature, classification, structure, methods of preparation, physical and chemical properties, uses,
identifcation of primary, secondary and tertiary amines.
Diazonium salts: Preparation, chemical reactions and importance in synthetic organic chemistry.
Biomolecules
Carbohydrates – Classification (aldoses and ketoses), monosaccahrides (glucose and fructose), oligosaccharides (sucrose, lactose, maltose), polysaccharides (starch, cellulose glycogen), importance.
Proteins – Elementary idea of ex- amino acids, peptide bond, polypeptides proteins, primary structure, secondary
structure, tertiary structure and quaternary structure (qualitative idea only), denaturation of proteins, enzymes.
Vitamins – Classification and functions.
Nucleic Acids : DNA & RNA.
Polymers :
Classification – natural and synthetic, methods of polymerization (addition and condensation), copolymerization. Some important polymers: natural and synthetic like polythene, nylon, polyesters, bakelite, rubber.
Chemistry in Everyday life:
1. Chemicals in medicines – analgesics, tranquilizers, antiseptics, disinfectants, antimicrobials, antifertility
dmgs, antibiotics, antacids, antihistamines.
2. Chemicals in food – preservatives, artifcial sweetening agents.
3. Cleansing agents – soaps and detergents, cleansing