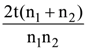JIPMER SOLVED PAPER-2019
Physics
1. Find the current in the 8Ω resistance in the given circuit.

(a) 2 A
(b) 3 A
(c) 4 A
(d) 5 A
2. In the given circuit find the heat produced between A and B (given C = 1 μF)

(a) 50 μJ
(b) 60 μJ
(c) 70 μJ
(d) 100 μJ
3. In the given circuit, if power rating of heater is 1000 Watt at 100 Volt, find the resistance R in figure so that heater produces power of 62.5 W.

(a) 5 Ω
(b) 7 Ω
(c) 10 Ω
(d) 8 Ω
4. Ba-122 has half life of 2 min. Experiment has to be done using Ba-122 and it takes 10 min to set up the experiment. If initially 80 g of Ba-122 was taken, how much Ba was left when experiment was started?
(a) 2.5 g
(b) 5 g
(c) 10 g
(d) 20 g
5. Find the value of θ in the given diagram.

(a) sin−1(2/√3)
(b) sin−1(1/√3)
(c) sin−1(1/2)
(d) sin−1(1/√2)
6. Find the position of final image from first lens. Given focal length of each lens is 10 cm.

(a) 40 cm
(b) 50 cm
(c) 45 cm
(d) 55 cm
7. If velocity of light in air is c, what will be the velocity of light in a medium of refractive index 1.4?
(a) 2.1 × 108 m/s
(b) 1.4 × 108 m/s
(c) 2.8 × 108 m/s
(d) None of these
8. If ratio of terminal velocity of two drops falling in air is 3 : 4, then what is the ratio of their surface area?
(a) 2/3
(b) 3/4
(c) 4/3
(d) 3/2
9. If frequency (v) of light is 5 × 106 Hz and speed of light in air is 3 × 108 m/s. Find the ratio of wavelength of light in a medium of refractive index ‘2’ to air
(a) 1/2
(b) 3
(c) 3/2
(d) 2
10. Find the maximum radius of circle so that the block can complete the circular motion.

(a) 5 cm
(b) 3 cm
(c) 2 cm
(d) 4 cm
11. Find the height of liquid in capillary tube, if surface tension of liquid = S, radius of capillary tube = r and acceleration due to gravity = g.
(a) ![]()
(b) ![]()
(c) ![]()
(d) None of these
12. Following circuit will act as

(a) NOR gate
(b) NAND gate
(c) AND gate
(d) OR gate
13. The rate of decrease of kinetic energy is 9.6 J/s. Find the magnitude of force acting on particle when it’s speed is 3 m/s.
(a) 3.2 N
(b) 4.8 N
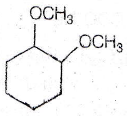
(c) 2.4 N
(d) 5.6 N
14. Maximum acceleration in SHM
(a) ω2A
(b) ω2A/2
(c) ω2A2
(d) 0
15. If frequencies are (v – 1), (v + 1), then find beats.
(a) 2
(b) 1
(c) 2
(d) 4
16. If mass (M), distance (L) and time (T) are fundamental quantities, then find dimension of torque.
(a) [ML2T−2]
(b) [MLT−2]
(c) [MLT]
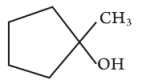
(d) [ML2T]
17. Find the correct diagram of electric lines of forces for negative charge.
(a) 
(b) 
(c) 
(d) 
18. A circuit contain two resistance R1 and R2 in series. Find the ratio of input voltage to voltage of R2.
(a) ![]()
(b) ![]()
(c) ![]()
(d) ![]()
19. Find the current drawn from the battery just after the switch is closed.

(a) E/2R
(b) 2E/R
(c) E/R
(d) 2E/3R
20. Find the average velocity when a particle complete the circle of radius 1m in 10 sec.
(a) 2 m/s
(b) 3.14 m/s
(c) 6.28 m/s
(d) zero
21. Speeds of a particle at 3rd and 8th sec are 20 m/s and zero respectively, then average acceleration between 3rd and 8th sec will be
(a) 3 m/s2
(b) 4 m/s2
(c) 5 m/s2
(d) 6 m/s2
22. If object starts from rest and covers angle of 60 rad in 10 sec in circular motion magnitude of angular acceleration will be
(a) 1.2 rad/sec2
(b) 1.5 rad/sec2
(c) 2 rad/sec2
(d) 2.5 rad/sec2
23. One object of mass 20 kg is moving with speed 10 m/s in west direction another object of mass 10 kg is moving with 15 m/s in North direction. Both collide and stick together. Choose the correct alternative.
(a) There K.E. is conserved as it is inelastic collision
(b) There K.E. is conserved on it is elastic collision
(c) There momentum is conserved as it is inelastic collision
(d) There momentum is conserved as it is elastic collision
24. One rod of length 2 m and thermal conductivity 50 unity is attached to another rod of length 1 m and thermal conductivity 100 unit. Temperature of free ends are 70°C and 50°C respectively. Then temperature of junction point will be
(a) 60°C
(b) 54°C
(c) 64°C
(d) 68°C
25. If three objects are thrown with velocity ![]() then find the ratio of their maximum heights.
then find the ratio of their maximum heights.
(a) 2 : 6 : 1
(b) 2 : 5 : 3
(c) 2 : 4 : 7
(d) 3 : 7 : 4
26. Find the distance between two consecutive nodes, if for a string T = 0.25 N, μ = 1 mg/m f = 250 Hz.
(a) 2m
(b) 1m
(c) 3m
(d) 1.5m
27. If escape velocity on earth surface is 11.1 km/h, then find the escape velocity on moon surface. If mass of moon is 1/81 times of mass of earth and radius of moon is 1/4 times radius of earth.
(a) 2.46 km/h
(b) 3.46 km/h
(c) 4.4 km/h
(d) None of these
28. Two disc having mass ratio 1/2 and diameter ratio 2/1 then find ratio of moment of inertia.
(a) 2 : 1
(b) 1 : 1
(c) 1 : 2
(d) 2 : 3
29. For a battery the electromotive force is 1.5 V. Terminal voltage is 1.25 V. Power supplied to external resistance is 2.5 W. Find the internal resistance of battery.
(a) 0.125 Ω
(b) 0.25 Ω
(c) 0.5 Ω
(d) 0.1 Ω
30. A projectile of mass 1 kg is projected with a speed of 10 m/s at an angle of 60° from the horizontal when projectile is at its highest point, its magnitude of angular momentum (about point of projection)
(a) ![]()
(b) ![]()
(c) 75 Nm/s
(d) 150 Nm/s
31. Two wire of same material having radius in ratio 2 : 1 and lengths in ratio 1 : 2. If same force is applied on them, then ratio of their change in length will be
(a) 1 : 1
(b) 1 : 2
(c) 1 : 4
(d) 1 : 8
32. If frequency of a photon is 6 × 1014 Hz, then find its wavelength.
[Take, speed of light, c = 3 × 108 m/s]
(a) 500 Å
(b) 500 nm
(c) 200 Å
(d) 200 nm
33. Find resonance frequency in the given circuit

(a) 1/√LC
(b) 2/√LC
(c) 1/2√LC
(d) 4/√LC
34. If a ball of mass 0.1 kg hits the ground from the height of 20 m and bounce back to the same height, then find out the force exerted on the ball if the time of impact is 0.04 sec.
(Take, g = 10 m/s2)
(a) ![]()
(b) ![]()
(c) ![]()
(d) ![]()
35. A galvanometer of 50 Ω resistance has 25 divisions. A current of 4 × 10−4 A gives a deflection of one division. To convert this galvanometer into a voltmeter having a range of 25 V, it should be connected with a resistance of
(a) 2500 Ω as a shunt
(b) 245 Ω as a shunt
(c) 2550 Ω in series
(d) 2450 Ω in series
36. A man applying force of 20 N on an object at an angle of 60° from the horizontal. If object moved by 20 m in horizontal direction, then work done by the force on the object is
(a) 300 J
(b) 400 J
(c) 100 J
(d) 200 J
37. What is dimensions of energy in terms of linear momentum (P), area (A) and time (T)
(a) [P1A1T1]
(b) [P2A2T−1]
(c) [P1A1/2T−1]
(d) [P1/2A1/2T−1]
38. Ratio of charge on positron to mass of positron is approximately
(a) +2 × 1011
(b) +5 × 1012
(c) −2 × 1011
(d) −5 × 1011
39. In the given circuit, find voltage across 12 Ω

(a) 12 Volt
(b) 36 Volt
(c) 72 Volt
(d) 48 Volt
40. Potential difference is given as
V(x) = −x2y volt. Find electric field at a point (1, 2)?
(a) 
(b) ![]()
(c) ![]()
(d) ![]()
41. A planet of radius R has a time period of revolution T. Find time period of planet of radius 9R?
(a) 3√3T
(b) 9T
(c) 27T
(d) 9√3T
42. A toy truck of mass 2m elastically collides with a toy car of mass m. speed of truck is v and car is at rest. Find the velocity of car after collision.
(a) 4v/3
(b) v/3
(c) v
(d) 2v/3
43. If resistivity of copper is 1.72 × 10−8 Ω-m and number of free electrons in copper is 8.5 × 1028/m3. Find the mobility.
(a) 4.25 × 10−3 m2/CΩ
(b) 6.8 × 10−3 m2/CΩ
(c) 8.5 × 10−3 m2/CΩ
(d) 3.4 × 10−3 m2/CΩ
44. Van De graff generator is used to
(a) create a high potential of range of few million volts
(b) create a low potential of range of few million volts
(c) to de-accelerate projectile; like protons, deuterons etc.
(d) it can not be used to study collision experiments in physics
45. Coercivity and retentivity of soft iron is
(a) high coercivity, high retentivity
(b) low coercivity, high retentivity
(c) low coercivity, low rententivity
(d) high coercivity, low retentivity
46. In YDSE angular fringe width is 0.1ʹ wavelength of light used is 6000 Å. Find the distance between two slits.
(a) 2 μm
(b) 3 μm
(c) 5 μm
(d) 6 μm
47. Lenz law is based on principle of conservation of
(a) Linear momentum
(b) Energy
(c) Charge
(d) Mass
48. Two objects of mass m each moving with speed u m/s collide at 90°, then final momentum is (assume collision is inelastic)
(a) mu
(b) 2 mu
(c) √2 mu
(d) 2√2 mu
49. A particle is projected at an angle of 30° from the horizontal with a speed of 10 m/s on the earth. It’s time of flight, range, velocity of impact are respectively T, R, V. If this projectile is projected on the moon then its time of flight, range and velocity of impact is (assume gravitational field on moon is g/6 m/s2)
(a) 6T, 6R, 6V
(b) 6T, 6R, V
(c) T, R, V
(d) T, 6R, 6V
50. A particle is moving with 10 m/s in a circle of radius 5m, find out magnitude of average velocity if particle moved by 60° in 1 sec.
(a) 5 m/s
(b) 10 m/s
(c) 5√3 m/s
(d) 20 m/s
51. A particle of mass 7 kg executing circular motion with time period of 11 sec. Find out centripetal force, if radius circle is 10 m.
(a) 30/7 N
(b) 40/7 N
(c) 30 N
(d) 160/7 N
52. A stationary wave equation is given as y = 20 sin(20x) cos (1000 t).
What will be the speed of stationary wave?
(a) 20 m/s
(b) 50 m/s
(c) 2 cm/s
(d) not defined
53. Huygen’s principle does not use
(a) Reflection
(b) Refraction
(c) Diffraction
(d) Point of spectra origin
54. When a light ray enters from oil to glass on oil-glass interface, then velocity of light changes by a factor of
[given, μoil = 2, μglass = 3/2]
(a) 4/3
(b) 3/4
(c) 3
(d) 1
55. For CE configuration n-p-n transistor.
Which of the following statement is correct?
(a) IC = IE + IB
(b) IB = IE + IC
(c) IE = IC + IB
(d) All of these
56. Psychrometer is used to measure
(a) relative humidity
(b) pressure
(c) temperature
(d) density
57. If same charge q is placed inside a sphere and cube having radius 1 m and side 2 m respectively. What will be the ratio of flux passing through them?
(a) 1 : 1
(b) 1 : 8
(c) 8 : 1
(d) 1 : 2
58. Induction furnace uses ……….. to produce heat.
(a) eddy current
(b) resistance
(c) capacitor
(d) None of these
59. The given combination will work as

(a) NAND gate
(b) OR gate
(c) AND gate
(d) NOR gate
60. In which radiation atomic number and mass number are not affected?
(a) α-radiation
(b) γ-radiation
(c) β-radiation
(d) None of these
Chemistry
61. ![]() Final product is
Final product is
(a) propene
(b) propanol
(c) cyclopropane
(d) propane-1, 2-diol
62. Which of the following can from H-bond?
(a) NH3
(b) R-CN
(c) R-O-R
(d) R-Br
63. Which of the following amino acid is optically inactive?
(a) Glycine
(b) Valine
(c) Alanine
(d) Histidine
64. In which of the following, oxidation state of phosphorus is +3?
(a) Orthophosphoric acid
(b) Pyrophosphoric acid
(c) Orthophosphorous acid
(d) Meta phosphoric acid
65. If ![]() react with Cl2 in presence of light and then react witih Na metal in dry ether, final product is
react with Cl2 in presence of light and then react witih Na metal in dry ether, final product is
(a) 
(b) 
(c) ![]()
(d) 
66. ![]()
![]()
What is suitable reagent (X)?
(a) H2 | Pt
(b) B2H6|THF, H2O2|OH−
(c) Br2 | HCl
(d) HBr | H2O2
67. What is the oxidation number of Cr in Na2Cr2O7?
(a) 2
(b) 6
(c) 10
(d) 16
68. What is the value of γ for mono atomic gas (ideal gas) ?
(a) 7/5
(b) 4/3
(c) 5/2
(d) None of these
69. Formula for half-life of a zero order reaction is
(a) C0/K
(b) C0/2K
(c) 2C0/K
(d) 2C0/2K
70. In an ideal gas equation which is constant?
(a) Temperature
(b) Pressure
(c) Volume
(d) Universal gas constant
71. If two atoms have equal number of electron, it is called
(a) isoelectronic
(b) isotone
(c) isobar
(d) None of these
72. Hund’s rule state that
(a) number of two e− can be in two separate orbitals
(b) number of two e− can be present with similar spin in a orbital
(c) no one e− can exist in d orbital
(d) None of the above
73. 19F−1, 16O−2 20Ne choose the correct statement
(a) Both O−2 and F− are isoelectronic
(b) All given have equal to of e−
(c) F− and Ne have equal number of e−
(d) All of the above
74. Which method is used to find halogen in organic compound?
(a) Duma’s method
(b) Leibig’s method
(c) Kjeldahl method
(d) Carius method
75. Which of the following is not iso electronic with H2S?
(a) F2 gas
(b) oxide ion
(c) Ca+2
(d) Sc+3
76. Number of σ and π bonds in C2H2 is respectively
(a) 2, 3
(b) 3, 2
(c) 4, 2
(d) 2, 4
77. If molarity of Cu+2 ions is 3 × 10−4 express this in ppm
(a) 0.3
(b) 0.2
(c) 0.1
(d) 0.6
78. Which of the following has octet around central atom?
(a) PF5
(b) SF6
(c) CCl4
(d) BF3
79. Which of the following is correct?
(a) SO32 ⇒ Tetrahedral
(b) NO2− ⇒ Trigonal planar
(c) ClO4− ⇒ Tetrahedral
(d) NH3 ⇒ Trigonal planar
80. If 50 ml of 0.1 HBr is mixed with 50 ml 0.2 M NaOH, find pH of resulting mixture
(a) 2.7
(b) 12.7
(c) 10.7
(d) 1.3
81. IUPAC name of  is
is
(a) 4-hydroxy-3-methyl cyclohexene
(b) 2-methyl cyclohex-3-en-1-ol
(c) 3-hydroxy-2-methyl cyclohexene
(d) 1-Hydroxy-3-methyl cyclohexene
82. Bisphenol A is
(a) Phenol + Ethanol
(b) Phenol + Aniline
(c) Phenol + Acetone
(d) Phenol + Ethanal
83. If equilibrium constant is 2.6 × 108 at 298 K, then find the value of ∆G°.
(a) −48.14 kJ
(b) −63.2 kJ
(c) −23.3 kJ
(d) +40 kJ
84. Which of the following is not correct?
(a) PhCH2Br > PhCHBrCH3 > PhCBr(CH3)2(SN1)
(b) R – I > R – Br > R – Cl (SN2)
(c) CH2 = CH – Cl < CH2 = CH – CH2 – Cl < PhCH2 – Cl (SN1)
(d) R – Cl < R – Br < R – I (SN1)
85. Invert sugar is mixture of
(a) D-glucose + D-fructose
(b) L-glucose + D-fructose
(c) L-glucose + D-glucose
(d) L-glucose + L-glucose
86. Which of the following is not allotrope of carbon?
(a) Diamond
(b) Fullerene
(c) Soot
(d) Graphite
87. Calculate molarity of one litre solution of 22.2 gm CaCl2.
(a) 0.4 M
(b) 0.2 M
(c) 0.8 M
(d) 0.6 M
88. If V = 1 litre, 10 mole of H2 and 10 mole of N2 gas are mixed temperature 26°C, then calculate pressure of the gas.
(a) 491 atm
(b) 300 atm
(c) 550 atm
(d) 600 atm
89. Which mixture is immiscible?
(a) C5H12 + Alcohol
(b) Alcohol + Amine
(c) H2O + Alcohol
(d) Alkane + Alkane
90. Silicon carbide is example of
(a) molecular solid
(b) metallic solid
(c) ionic crystal
(d) covalent solid
91. Which type of defect present in NaCl?
(a) Frenkel defect
(b) Schottky defect
(c) F-centre defect
(d) None of these
92. Which one follow 18 electron octet rule?
(a) [Mn(CO)5]
(b) [Cr(CO)5]
(c) [Fe(CO)5]
(d) None of these
93. ![]()
Suitable reagent for conversion of following reaction?
(a) NaBH4
(b) Ni/H2
(c) Zn/Hg/HCl
(d) Red P + Hi
94. Bordeaux mixture consist of
(a) CuSO4 + Ca(OH)2
(b) CuSO4 + CaCl2
(c) ZnSO4 + Mg(OH)2
(d) FeSO4 + Ba(OH)2
95. BrO3− changes into Br2 in an acidic medium of a unbalanced equation. How many electron should be present on the balanced equation?
(a) 10 electron in left
(b) 6 electron in left
(c) 3 electron in left
(d) 3 electron in left
96. Frenkel defect is present in which of the following?
(a) NaOH
(b) NaI
(c) AgBr
(d) None of these
97. Butter is a colloidal solution of
(a) solid-solid
(b) liquid-solid
(c) solid-liquid
(d) gas-solid
98. Azimuthal quantum number (ℓ) defined
(a) shape of orbitals
(b) orientation of orbitals
(c) energy of orbitals
(d) size of orbitals
99. 2KHCO3 →… + CO2 + H2O find amount of gases formed (in lit). When amount of KHCO3 is 33 gm.
(a) 5.6
(b) 11.2
(c) 7.46
(d) 22.4
100. ![]() final product C is
final product C is
(a) CH3CH2OH
(b) CH3CH2 – Br
(c) CH3 – CH2 – I
(d) 
101. Glucose ![]() IUPAC name of Y and Z
IUPAC name of Y and Z
(a) Hexa hydroxyl heptanoic acid, heptane
(b) Hepta hydroxyl hexanoic acid, hexane
(c) Penta hydroxyl hexanoic acid, hexane
(d) Hepta hydroxyl hexanoic acid, heptane
102. Write the IUPAC name of given structure

(a) Para bromo benzene
(b) 1, 4-di bromo benzene
(c) Both (a) and (b)
(d) Meta bromo benzene
103. Which of the following statement is correct for isotope of carbon?
(a) graphite is conductor of electricity
(b) diamond have all sp3 carbon
(c) graphite is more stable thermodynamically than diamond
(d) All are correct
104. Write IUPAC name of the following

(a) 2-bromo-2-ethyl hexanal
(b) 3-bromo-2-methyl hexanal
(c) 2-methyl-3-bromo hexanal
(d) 3-bromo-2-formyl hexane
105. Most common isotopes of hydrogen (non-radioactive) is
(a) protium
(b) deuterium
(c) tirium
(d) All of these
106. How many ions obtain after dissociation of this complex [Co(NH3)6]Cl3?
(a) 3
(b) 2
(c) 5
(d) 4
107. Given, ![]() The potential for the cell
The potential for the cell
Cr|Cr3+(0.1M) || Fe2+(0.01 M)Fe is
(a) −0.339 V
(b) −0.026 V
(c) 0.26 V
(d) 0.3 V
108. The magnetic moment of Ni2+ ion (atomic number of Ni = 28) in BM unit is
(a) 1.73
(b) 4.81
(c) 5.96
(d) 2.84
109. Which one of the following nitro compound does not react with nitrous acid?
(a) ![]()
(b) 
(c) 
(d) 
110. The main product obtained from phenol with PCl5 is
(a) BHC
(b) hexachlorobenzene
(c) chlorobenzene
(d) triphenyl phosphate
111. An element with mass number 81 contains 31.7% more neutrons as compared to protons. It symbol is
(a) 40X81
(b) 35X81
(c) 81X317
(d) 40X81
112. Which of the following will not be formed when calcium acetate is distilled with calcium formate?
(a) Acetone
(b) Propanal
(c) Ethanal
(d) Methanal
113. If half-life of a surface is 36 minutes. Find amount left after 2 hrs. Initial amount is 10 gm?
(a) 1 g
(b) 2 g
(c) 3 g
(d) 4 g
114. Moles of N2 = 0.4 and moles of O2 = 0.1 find ![]() (partial pressure) N2 = ? at atmospherim pressure
(partial pressure) N2 = ? at atmospherim pressure
(a) 0.2 atm
(b) 0.8 atm
(c) 0.6 atm
(d) 0.4 atm
115. What is hybridization of O in H2O?
(a) sp
(b) sp3
(c) sp2
(d) No hybridization
116. Which of the following act as epimeric pair?
(a) Glucose and fructose
(b) Fructose and mannose
(c) Glucose and mannose
(d) Glucose and sucrose
117. Which can not behave as nucleophile for SN2 reaction?
(a) H2O
(b) CN−
(c) NH2−
(d) I−
118. Which of the following rate gas is the abundant in air?
(a) He
(b) Ne
(c) Ar
(d) Kr
119. Saccharin an artificial sweetener is manufactured form
(a) toluene
(b) cyclohexane
(c) starch
(d) cellulose
120. During nitration of benzene the attacking electrophile is
(a) NO3−
(b) NO2−
(c) NO2+
(d) HNO3
Biology
121. Verticillaster inflorescence is found in
(a) mustard
(b) Ocimum
(c) China rose
(d) sunflower
122. Zooidogamy is found in
(a) Cedrus
(b) Pinus
(c) Cycas
(d) Both (b) and (c)
123. Dark reaction
(a) in light
(b) in dark
(c) requires product of light reaction
(d) All of the above
124. Vernalisation is
(a) low pH treatment
(b) low temperature treatment
(c) high temperature treatment
(d) high pH treatment
125. An interaction in which one organism is benefitted and other is unaffected
(a) predation
(b) commensalism
(c) mutualism
(d) parasitism
126. Karyotype of Down’s syndrome has how many chromosomes?
(a) 43
(b) 46
(c) 47
(d) 45
127. Hardest substance in plant kingdom is
(a) saple
(b) corolla
(c) sporopollenin
(d) anther
128. Which of the following is not a micronutrient?
(a) B
(b) Zn
(c) Mn
(d) Mg
129. Cell placed in hypertonic solution is shown by
(a) 
(b) 
(c) 
(d) Both (a) and (b)
130. Genetic variation cannot be effectively brought about by
(a) transduction
(b) mutation
(c) transformation
(d) amplification
131. Which of the following is not considered as a secondary messenger?
(a) Acetylcholine
(b) cAMP
(c) Ca2+
(d) diacylglycerol
132. Which is also called molecular glue
(a) DNA gyrase
(b) DNA helicase
(c) DNA ligase
(d) DNA polymerase
133. In DNA 20% bases are adenine. What percentage of bases are pyrimidines?
(a) 30%
(b) 60%
(c) 50%
(d) 20%
134. Most advanced invertebrates are
(a) Arthropoda
(b) Annelida
(c) Mollusca
(d) Cephalopoda
135. Gastrulation means
(a) conversion of blastula into morula
(b) formation of 3 germs layers
(c) a phase in which organogenesis takes places
(d) a phase characterized by inner cell mass
136. Traditional anesthesia is obtained from
(a) Datura
(b) poppy
(c) Cannabis
(d) erythroxylum
137. Band 3 protein in RBC has the function of
(a) it mediates the exchange of cellular HCO3− with Cl− in plasma
(b) it is a cytoplasmic protein
(c) it mediates the exchange of cations between cell and plasma
(d) Both (a) and (b)
138. Hypothalamus can regulate
(a) anger
(b) patience
(c) thermoregulation
(d) pleasure
139. Choose correct option for larva of housefly.
(a) Grub
(b) Cypris
(c) Maggot
(d) Wriggler
140. Chromosome walking is
(a) used in genetic mapping
(b) observed during mitosis
(c) to see position of ribosomes on nuclear membrane
(d) to check the expression of mutant genes
141. Which one is only found in plant?
(a) Ribosome
(b) Mitochondria
(c) Glyoxysomes
(d) Lysosome
142. Kranz anatomy is found in
(a) C3-plants
(b) Only in moncots
(c) Both C4 and C3-plants
(d) C4-plants
143. What will be the DNA content and number of chromosomes in a cell of after S-phase as compared to the gamete of the same organism?
(a) Same DNA content but double chromosome number
(b) Four times DNA content but double chromosome number
(c) Same DNA content but half chromosome number
(d) Half DNA content but double chromosome number
144. Radula is a part of which animal.
(a) Mollusca
(b) Poriferans
(c) Coelenterata
(d) Annelida
145. Chordae tendineae is found in
(a) heart of cockroach
(b) heart of Amphibians
(c) heart of Reptiles
(d) heart of Mammals
146. Length of oesophagus is
(a) 25 cm
(b) 55 cm
(c) 33 cm
(d) 45 cm
147. Citrulline structure
(a) 
(b) 
(c) 
(d) 
148. Bradykinin is a
(a) antibody
(b) hormone
(c) enzyme
(d) receptor
149. Reproduction without actual fertilization is called as
(a) parthenogenesis
(b) parthenocarpy
(c) pseudo-reproduction
(d) All of these
150. Coacervates were discovered by
(a) Huxley
(b) Fox
(c) Oparin
(d) Miller
151. Which of the following is not a stop codon?
(a) UAA
(b) UAC
(c) UAG
(d) UGA
152. Which of the following is uricotelic?
(a) Insects
(b) Birds
(c) Lizards
(d) All of these
153. To make chromosomal studies easier, chromosomes are classified into certain groups. So, the chromosome number 21, 22, and Y are listed in
(a) A
(b) D
(c) E
(d) G
154. Choose the incorrect about gemmules.
(a) They resist dessication
(b) They are internal buds
(c) They are asexual structure which are produce by binary fission
(d) They can give rise to new organisms
155. Choose the correct regarding antibodies.
(a) IgA – Helps in allergic reaction
(b) IgG – Cross placenta
(c) IgE – Found in secretions
(d) IgM – Exists as dimer
156. During DNA replication, supercoilling is relaxed by
(a) primase
(b) polymerase
(c) DNA topoisomerase
(d) SSBPs
157. Structure analogous to the placenta in mammals.
(a) Chorion
(b) Amnion
(c) Yolk sac
(d) Allantois
158. In annealing
(a) primers anneal to the DNA template
(b) Taq polymerase adds nucleotides to the annealed primer
(c) two strands of the DNA separate
(d) temperature is 92°C
159. Which of the following is not true?
(a) β – galactosidase – lac operon
(b) Photorespiration – C3 – plants
(c) Krebs cycle – mitochondrial matrix
(d) Sothern hybridization – related to identification of RNA sequence
160. Carcinogen present in cigarette smoke is
(a) benzopyrene
(b) nicotine
(c) carbon monoxide
(d) All of these
161. Atlas is
(a) 1st cervical vertebrae
(b) 2nd cervical vertebrae
(c) 1st thoracic vertebrae
(d) 2nd lumbar vertebrae
162. Typhoid is caused by
(a) penumonia
(b) filarial worm
(c) Salmonella typhii
(d) None of the above
163. Chl-b differ from chl-a by
(a) −CH3 group
(b) CHO group
(c) COOH group
(d) CH2OH group
164. Number of chromosomes present in secondary spermatocyte is
(a) 22
(b) 23
(c) 24
(d) 25
165. Flame cells are present in
(a) Aschelminthes
(b) Platyhelminthes
(c) Annelida
(d) Cephalochordata
166. Which one of the labeled part utilizes fructose as a source of energy?

(a) Head
(b) Tail
(c) Acrosome
(d) Middle piece
167. A normal man and a normal woman both having one of their parent albino, if get married then what is the probability of their child of being albino?
(a) 20%
(b) 25%
(c) 75%
(d) 80%
168. Which is the correct sequence of modification and transport of secretory vesicles?
(a) RER → Cell membrane → Secretory vesicles → Golgi body
(b) RER → Secretory vesicles → Cell membrane
(c) RER → Cell membrane → Secretory vesicles → Golgi body
(d) SER → Golgi body → Cell membrane → Secretory vesicles
169. Among the following choose the correct epimers.
(a) Glucose, galactose
(b) Glucose, fructose
(c) Glucose, ribose
(d) Gluconic acid, glucose
170. Gynaecomastia is present in which syndrome?
(a) Turner
(b) Klinefelter
(c) Down
(d) None of the above
171. In gluconeogenesis, which of the following cannot act as substrate?
(a) Fructose
(b) Palmitic acid
(c) Glycine
(d) Glycerol
172. Identify the diagram of heterocyst.
(a) 
(b) 
(c) 
(d) 
173. Level of LH is maximum
(a) just before ovulation
(b) just after ovulation
(c) at the time of ovulation
(d) during menstrual bleeding phase
174. Number of chromosomes present in spermatogonium is
(a) 46
(b) 23
(c) 48
(d) 43
175. Which one of them is odd with respect to human male?
(a) absence of one X-chromosome
(b) addition of one X-chromosome
(c) presence of XY-chromosome
(d) absence of Barr body
176. Eutrophication is caused due to
(a) accumulation of minerals
(b) effect of UV-C
(c) accumulation of metals only
(d) accumulation of zooplankton
177. Synaptonemal complex is formed
(a) during anaphase
(b) during metaphase
(c) during prophase-II
(d) during prophase-I of meiosis
178. Which one is the oldest in following?
(a) Gymnosperms
(b) Angiosperms
(c) Bryophytes
(d) None of these
179. RNA binds to mRNA through
(a) anticodon loop
(b) T Ψ C loop
(c) amino acid binding loop
(d) D loop
180. Find odd one out.
(a) Stamen
(b) Stigma
(c) Style
(d) Ovary
English and Quantitative Reasoning
Directions (Q. Nos. 181-185) Read the following passage carefully and answer the questions given below.
In this passage, Ria and Gargi give their opinions about a new painting by Marcello Vasquez. Ria, ‘The new painting is spectacular! It is certainly the best work ever completed by Marcello Vasquez. I like the new painting for several reasons. First, the painting is large. It almost covers the entire wall! Second, the painting is inexpensive. Normally, a painting like this would cost thousands of dollars. But this painting costs less than $500. Third, the painting is colourful. Red and green dominate the canvas, peppered with bright spots of yellow.’
Gargi, ‘The new painting is horrendous! Marcello Vasquez should be ashamed of his work. I haven’t seen a painting this bad in a long time. I dislike the new painiting for several reasons. First, the painting is far too large. It almost covers the entire wall! I think small paintings are better than large ones. Second, the painting is inexpensive. This tells me that Vasquez must have been unable to sell the painting for a higher price. Now, he is desperate. As a result, it appears he is trying to give the painting away. Third the painting is colourful. Although I usually like a painting to be colourful. Although I usually like a painting to be colourful, I strongly dislike the colours Vasquez uses. To p u tint plainly, the combination of red, green and yellow is ugly.’
181. With respect to how they like new painting Ria and Gargi?
(a) agree
(b) strongly agree
(c) disagree
(d) strongle disagree
182. Ria and Gargi both view the new painting as
I. Large II. Inexpensive III. Colourful
Select the correct answer using the codes given below
(a) Only I
(b) I and II
(c) II and III
(d) All of these
183. In the second paragraph Gargi said it can be understood that ‘desperate’ belongs to which of the following word groups?
(a) Passionate, enthusiastic, excited
(b) Idiotic, silly, foolish
(c) Anxious, worried, troubled
(d) Improverished, broke poor
184. As used in the passage, which of the following words always has/have negative meaning?
I. Spectacular II. Horrendous IIII. Inexpensive
Select the correct answer using the codes given below
(a) Only I
(b) Only II
(c) II and III
(d) All of these
185. Gargi and Ria go to see another painting. The painting is small, expensive and colourful, who would probably like this painting?
(a) Ria
(b) Gargi
(c) Both Ria and Gargi
(d) Neither Ria nor Gargi
Directions (Q. Nos. 186-187) Each of the following question consist of a word, followed by four words or group of words. Select the antonym of the word given below.
186. Expansion
(a) Depression
(b) Negligent
(c) Precise
(d) Enlargement
187. Thrift
(a) Parsimony
(b) Dismantle
(c) Prodigality
(d) Essential
Directions (Q. Nos. 188-190) Choose the correct option out of the choices given below.
188. He will abide ………. his friend Suresh in all circumstances.
(a) with
(b) by
(c) along
(d) to
189. He should not be blind ………….. the misdeeds of his son Rahul.
(a) at
(b) to
(c) with
(d) on
190. God had destine him ………. the post of Prime Minister of India
(a) for
(b) to
(c) with
(d) on
191. In a certain code language, ‘SAFER’ is written as ‘5@3#2’ and ‘RIDE’ is written as ‘2©%#’, how would ‘FEDS’ be written in that code?
(a) 3#©5
(b) 3@%5
(c) 3#%5
(d) 3#%2
192. Find the missing number from the given response.

(a) 72
(b) 720
(c) 7200
(d) 38
193. If the first and second letters in the word DEPRESSION were interchanged, also the third and fourth letters, the fifth and the sixth letters and so on, then which of the following would be seventh letter from the right.
(a) O
(b) P
(c) R
(d) S
194. Today is Thursday. The day after 59 days will be
(a) Sunday
(b) Monday
(c) Tuesday
(d) Wednesday
195. Which of the following represents coal mines, factories and fields?
(a) 
(b) 
(c)
(d)
196. Find out the missing term in the series.
1, 8, 27, ?, 125, 216
(a) 52
(b) 58
(c) 64
(d) 65
197. If ‘+’ means ‘×’, ‘−’ means’ ‘+’, ‘x’ means ‘÷’ and ‘÷’means ‘−’, then 6 – 9 + 8 × 3
(a) −2
(b) 6
(c) 10
(d) 12
198. What is the water image of

(a)
(b) 
(c)
(d)
199. A piece of paper is folded and punched as shown in the figure below.

How will it appear when unfolded.

200. Here are some words translated from an artificial language. Mallon piml means blue light mallon tifl means blue berry arpan tifl means rasp berry?
Which word could means ‘light house’?
(a) tiflmallon
(b) pimlarpan
(c) mallonarpan
(d) pimldoken



















































 will be proportional to
will be proportional to






 where y and x are measured in metre. Which of the following statements is true?
where y and x are measured in metre. Which of the following statements is true?











 the earlier value
the earlier value


 with the horizontal, it reaches maximum height H2. The relation between the horizontal range R of the projectile, H1 and H2 is
with the horizontal, it reaches maximum height H2. The relation between the horizontal range R of the projectile, H1 and H2 is



























































































































































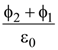




































 The current produced due to the motion of electron is
The current produced due to the motion of electron is