| Go to all OUAT Previous Year Question Papers | 2022, 2021, 2020, 2019, 2018, 2017, 2016, 2015, 2014, 2013, 2012,2011, 2010, 2009, 2008, 2007 |
OUAT Previous Question Paper-2022
PHYSICS
Questions : 66
1. If a heating device is able to melt 2000 gm of ice at −5°C in 10 min, then its power expressed in horse power will be-
(Specific heat of ice = 0.5 cal g−1 °C−1, latent heat of fusion of ice is 80 cal/g).
(A) 368 hp
(B) 0.368 hp
(C) 1.55 hp
(D) 1155 hp
2. The body of mass ‘m’ is rotating about a fixed axis with the radius of the path ‘r’ and linear velocity ‘v’. Its angular momentum is :
(A) mr/v
(B) mvr2
(C) mvr
(D) mv/r
3. The number of significant figures in the following : 5.700 m and 0.006400 kg are, respectively, _____.
(A) 4, 2
(B) 4, 4
(C) 2, 2
(D) 2, 4
4. If both the charge and electric field on the capacitor is doubled, then its capacitance :
(A) becomes double
(B) remains the same
(C) becomes four times
(D) becomes half
5. Match Column A with Column B
(A) p → ii, q → iii and r → i
(B) p → iii, q → i and r → ii
(C) p → i, q → iii and r → i
(D) p → ii, q → i and r → iii
6. A free neutron, unlike a free proton, is unstable. It decays into :
(A) an electron and an antineutrino
(B) a proton and antineutrino
(C) a proton, an electron and an antineutrino
(D) a proton and an electron
7. If a plane electromagnetic wave propagating in space has an electric field of amplitude 2.7 × 105 V/m, then the amplitude of the magnetic field is :
(A) 3 × 10−4 T
(B) 3 × 10−5 T
(C) 9 × 10−5 T
(D) 9 × 10−4 T
8. Inertial Mass is _____ and Gravitational Mass is _____.
(A) The measure of inertia and it follows from Newton’s second law of motion, the measure of gravitational pull and it follows from Newton’s law of Gravitation.
(B) The measure of gravitational pull and it follows from Newton’s law of Gravitation, the measure of inertia and it follows from Newton’s second law of motion.
(C) The measure of gravitational pull and it follows from Newton’s second law of motion; the measure of inertia and it follows from Newton’s law of Gravitation.
(D) The measure of inertia and it follows from Newton’s law of Gravitation; the measure of gravitational pull and it follows from Newton’s second law of motion.
9. For unit magnification, the distance of an object from a concave mirror of focal length 10 cm will be :
(A) −20 cm
(B) 20 cm
(C) −10 cm
(D) 10 cm
10. According to the third law of thermodynamics:
(A) it is possible to reach 0 K with an ideal refrigerator
(B) it is possible to reach 0°C in any physical process.
(C) it is impossible to reach 0°C with an ideal refrigerator
(D) it is impossible to reach 0 K in any physical process.
11. Ohm’s law is NOT applicable in the case of which of the following electrical equipment?
(A) Filament of a light bulb
(B) Fan motor
(C) All ohmic conductors
(D) Resistors
12. Match Column A with Column B
(A) p-ii, ,q-iv,, r-i and s-iii
(B) p-iii, ,q-iv,, r-i and s-ii
(C) p-iii, ,q-i,, r-iv and s-ii
(D) p-ii, ,q-i,, r-iv and s-iii
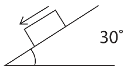
13. The dimension of the product of surface tension and length is :
(A) [M L−1 T−1]
(B) [M L T−2]
(C) [M L T−1]
(D) [M L0 T−2]
14. In an electromagnetic wave, the electric and magnetic fields are 200 V m−1 and 0.426 A m−1. The maximum energy flow is :
(A) 185.2 W/m2
(B) 45.2 W/m2
(C) 850.2 W/m2
(D) 85.2 W/m2
15. A 0.5 kg block is pushed against a wall with a horizontal force of 10 N. If the coefficient of static and kinetic friction between the wall and the block are 0.5 and 0.3, respectively, then :
(Take g = 10 m/sec2.)
(A) the frictional force on the block is 4.0 N
(B) the block moves down with an acceleration.
(C) the frictional force on the block is 5.0 N
(D) the normal force on the block is 5.0 N
16. In instrinsic semiconductor, the number of free electrons, ne and number of holes, nh are related as :
(A) ne < nh
(B) ne = nh
(C) ne > nh
(D) ne = nh = 0
17. The slope of stress vs. strain graph for a wire subjected to increasing load, upto proportionality limit gives :
(A) Poisson’s ratio of the wire
(B) Rigidity modulus of the wire
(C) Young’s modulus of the wire
(D) Bulk modulus of the wire
18. If the cabin of an ideal refrigerator is at −4°C, then for its coefficient of performance to be 5, the outside temperature should be nearly _____ °C (Choose the closest value)
(A) −20
(B) 323
(C) 52
(D) 27
19. Consider a tightly wounded 40 turn coil of radius 8 cm, carrying a current of 1 A. The magnitude of the magnetic field at the centre of the coil will be :
(A) 1.57 G
(B) 3.14 G
(C) 12.56 G
(D) 6.28 G
20. In which of the following cases the force acting on an object is one newton?
(a) Object of 1 kg moving with a constant velocity of 1 m/sec.
(b) Object of 1 g moving with a constant acceleration 1 m/sec2.
(c) Object of 1 kg moving with a constant acceleration 1 cm/sec2.
(d) Object of 1 kg moving with a constant
(A) (a)
(B) (b)
(C) (c)
(D) (d)
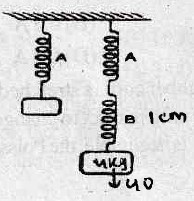
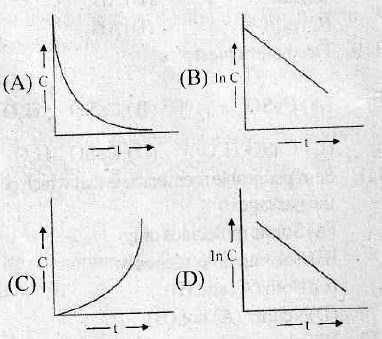
21. In an arrangement of two rods A and B joined in series, rod A is the upper rod and is of length 2l and radius of cross section r. Rod B is the lower rod and is of length l and radius of cross section 2r. If the upper end of rod A is fixed to a rigid roof and the lower end of rod B is given a twist of θ°, the angle of twist at the joint of the two rods A and B is _____.
22. A particle starts from (0m, 0m) at t = 0 s with a velocity of and moves in x-y plane with a constant acceleration of
Here symbols have their usual At what time is the x coordinate of the particle 27 m ?
(A) 2.0 s
(B) 4.0 s
(C) 3.0 s
(D) 1.0 s
23. In an enclosure two gases A and B, with densities in the ratio 18 units and 44 units are mixed. The volume of gas A is one part and gas B is three part of the total volume of the enclosure. Assuming that they do not react chemically, the speed of sound in gas A and B is the speed of sound in this mixture is 340 m/sec will be___.
(A) 314 m/sec and 419 m/sec
(B) 419 m/sec and 314 m/sec
(C) 708 m/sec and 289 m/sec
(D) 289 m/sec and 708 m/sec
24. Match Column A with Column B
(O is Optical Centre, F is first Principal Focus)
(A) p-ii, q-iv, r-i and s-iii
(B) p-iii, q-iv, r-i, and s-ii
(C) p-ii, q-i, r-iv and s-iii
(D) p-iii, q-i, r-iv and s-ii
25. If the force is acting along-X direction on a body moving along +X direction, then work done here :
(A) does not depends on direction
(B) is zero
(C) is negative
(D) is positive
26. The linear velocity of the tip of the second hand in a given all clock is : (Length of the second had is 10 cm.)
(A) 0.001 m/sec
(B) 0.1 m/sec
(C) 10 m/sec
(D) 0.01 m/sec
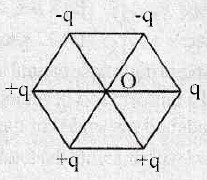
27. Infinitely large number of point charges, each equal to q are placed at positions x = 1, 2, 4, 8….. Calculate the electrostatic potential at the origin.
(A) πε0/q
(B) q/2πε0
(C) 2q/πε0
(D) q/3πε0
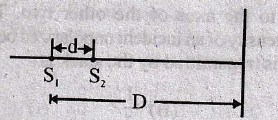
28. Two wires X and Y are placed parallel to each other at a distance of d carrying the same current in the same direction. At what distance between them, the magnetic field will be zero?
(A) d/4
(B) 1/d
(C) d
(D) d/2
29. There are two clocks. The first one works on the basis of oscillations of simple pendulum and the other works on the oscillation of a spring mass system on Earth. If these two are now taken to Pluto, then :
(A) the pendulum clock beats the same time as on Earth and the spring mass system does not beat same time as on Earth.
(B) The pendulum clock does not beat the same time as on Earth and the spring mass system does not beat the same time as on Earth.
(C) The pendulum clock beats the same time as on Earth and the spring mass system beats the same time as on Earth.
(D) The pendulum clock does not beat the same time as on Earth and spring mass system beats the same time as on Earth.
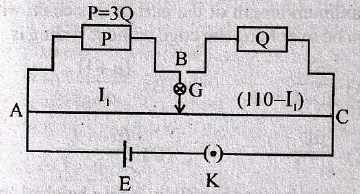
30. A coil having 450Ω resistance is placed in a magnetic field that varies with time t (sec) as ϕ = 45t2 + 2t. The current in the coil at t = 2 sec is :
(A) 0.404 A
(B) 0.54 A
(C) 0.5 A
(D) 0.32 A
31. SI unit and dimensions of activity of a radio-active sample is :
(A) S−1, [T−1]
(B) H2, [T−1]
(C) S, [T]
(D) Bq, [T−1]
32. For a body executing SHM the distance between the positive and negative extreme is 0.5 m and if it oscillates with a frequency of 40 hertz, the equation for its displacement (y) is ______.
(A) y = 0.25 sin (40t)
(B) y = 0.5 sin (40t)
(C) y = 0.25 sin (80πt)
(D) y = 0.5 sin (80 πt)
33. If the battery and galvanometer positions get interchanged in a Wheatstone bridge, the deflection in the galvanometer will :
(A) change to lower value
(B) change to higher value
(C) not change
(D) move in either direction
34. In an isothermal process, if the volume of 2 moles of a monoatomic gas (Y = 1.4) is explanded from V to 10V at a temperature 1°C, the amount of work done is ____.
(A) 4.6 RT loge10
(B) 4.6 RT
(C) 2.3 RT loge10
(D) 2.3 RT
35. On what following factors does Young’s Modulus of a given wire depend on ?
(i) Length
(ii) Area of cross-section
(iii) Tension in the wire
(iv) Material of the wire
(A) Only (ii) and (iii)
(B) Only (ii) and (iv)
(C) Only (iv)
(D) Only (i) and (iv)
36. Lorentz force is the :
(A) force of attraction or repulsion between two like and unlike charges, respectively.
(B) force on a point charge moving in presence of both the electric field and the magnetic field.
(C) force on a point charge at rest in the presence of both the electric field and the magnetic field.
(D) force that acts between the protons and neutrons of atoms.
37. Select the INCORRECT statement regarding electromagnetic waves.
(A) In free space they travel at the speed of light.
(B) They are transverse in nature.
(C) Their speed is same in all mediums.
(D) They are produced by accelerating charges.
38. The position of a particle is given by
Where r is in meters and t in seconds, i, j and k are unit vectors along x-axis, y-axis and z-axis, respectively. The magnitude of the velocity of the particle at t = 0.5 s is :
(A) 6.0 m/sec
(B) 3.0 m/sec
(C) 4.0 m/sec
(D) 5.0 m/sec
39. The eddy current magnitude does not depend upon the :
(A) magnetic flux
(B) magnetic field
(C) electric field
(D) resistance of conductor
40. The charge of two-point charges halved and the distance between them made four times, the force between them :
(A) remains the same
(B) gets double
(C) gets four times
(D) gets 1/64 times
41. The capacitance of a capacitor depends on the
(A) potential of conductors
(B) shape of conductors
(C) charge of conductors
(D) charge of densities
42. Change in kinetic energy of a body of mass 1kg moving along a circular path with the constant speed 10 m/s is :
(A) 10 J
(B) 0 J
(C) 1 J
(D) 5 J
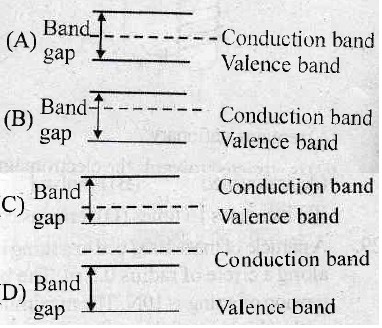
43. Materials A, B and C have energy band gap (Eg) of zero, < 3eV and > 3eV, respectively. Materials A, B and C can be classified as :
(A) Semiconductor, insulator and metal
(B) Metal, conductors and insulator
(C) Insulator, metal and semiconductor
(D) Metal, semiconductor and insulator
44. If an organ pipe closed at one end is suddenly opened, the fundamental frequency in the open pipe formed is found to be increased by 50 Hz. Then the 3rd overtone frequency of the closed pipe is _____.
(A) 350 Hz
(B) 250 Hz
(C) 150 Hz
(D) 50 Hz
45. An object 10m away from a concave mirror of focal length 1m, starts moving at a speed of 5m/sec towards the mirror. The average speed of the image is approximately :
(A) 1/7 m/sec
(B) 5 m/sec
(C) 1/5 m/sec
(D) 7 m/sec
46. Which of the following statement(s) is/are correct about errors in measurements?
(a) Imperfections in experimental procedures cause random errors.
(b) Unpredictable fluctuations in temperature cause systematic errors.
(A) Both (a) and (b)
(B) Only (a)
(C) Neither (a) nor (b)
(D) Only (b)
47. If a planet sweeps 2 lakh km2 are in the month of February 2022, then the area swept by it in one week of any month in any year will be ______.
(A) 5 × 106km2
(B) 8 × 1010km2
(C) 5 × 1010km2
(D) 8 × 106km2
48. Two circular metal plates with identical dimensions are rotating about an axis perpendicular to the plane of the body and passing through the centre. If mass of the first plate is double the mass of the second plate, then their moments of inertia are in the ratio :
(A) 4 : 1
(B) 1 : 2
(C) 1 : 1
(D) 2 : 1
49. The difference between the reciprocals of focal length and image distance is equal to the ______ in a spherical mirror.
(A) radius of curvature
(B) object distance
(C) Inverse of object distance
(D) Inverse of radius of curvature
50. Two coils are placed close to each other. The mutual inductance of the pair of coils depends upon :
(I) the relative position and orientation of the two coils
(II) the cross-section area of the wires of the coils.
(III) the current changing rates of both the coils.
Which of the above statement is/are true?
(A) I and II
(B) II and III
(C) Only III
(D) Only I
51. In two different places on earth, A and B, simple pendulum experiment was conducted with the same pendulum. It is found that in place A, 20 oscillations take 97.8 sec and at place B, 20 oscillations take 100 sec. The ratio of accelerations due to gravity of place B to place A is nearly____.
(A) 0.95
(B) 0.97
(C) 1.02
(D) 1.46
52. If 3 moles of diatomic gas are made to dissociate into a monotomic gas at room temperature, then the change in its degrees of freedom will be _____.
(A) 1.8 × 1024
(B) 1.2 × 1024
(C) 0.6 × 1024
(D) 2.4 × 1024
53. Consider the velocity (v) and acceleration (a) of a ball thrown vertically upward. During its motion upwards (downward direction taken to be negative) :
(A) v is positive and a is negative
(B) both v and are positive
(C) v is negative and a is positive
(D) both v and a are negative
54. Match Column A with Column B.
(A) p-iii, q-iv, r-i and s-ii
(B) p-ii, q-iv, r-i and s-iii
(C) p-ii, q-i, r-iv and s-iii
(D) p-iii, q-i, r-iv and s-ii
55. Magnetic flux is defined as :
(A) the total number of magnetic filed lines passing through a given area and perpendicular to it.
(B) the total current through a plane
(C) the total charge per unit volume
(D) the total EMF in a closed loop
56. For same increase in volume if WP, WT, WQ are works done in Isobaric, Isothermal and Adiabatic process, ascending order of these works done is _____.
(A) WQ < WP < WT
(B) WQ < WT < WP
(C) WT < WP < WQ
(D) WP < WT < WQ
57. Among the following thermodynamic quantities, the point functions are (i) __ and path functions are (ii) ___.
(a) external work (b) internal energy
(c) heat supplied (d) entropy
(A) (i) (a), (b) (ii) (c), (d)
(B) (i) (a), (d) (ii) (b), (d)
(C) (i) (b), (d) (ii) (a), (c)
(D) (i) (a), (c) (ii) (b), (d)
58. A fruit falling from the tree is under the action of gravitational force. Here force is :
(A) viscous force
(B) non-conservative force
(C) Elastic force
(D) Conservative force
59. Match Column A with Column B
(A) p-ii, q-i, r-iv and s-iii
(B) p-iii, q-iv, r-i and s-ii
(C) p-ii, q-iv, r-i and s-iii
(D) p-iii, q-i, r-iv and s-ii
60. A particle is in equilibrium under the action of three concurrent forces F1, F2 and F3 are given by :
F2 = (2.0 N)i + (3.0 N)j + (4.0 N)k
F3 = (3.0 N)i – (4.0 N)j – (5.0 N)k
Then F1 is :
(A) (1.0N)i – (7.0 N)j – (9.0N)k
(B) −(5.0N)i + (1.0 N)j + (3.0N)k
(C) −(2.0N)i + (7.0N)j + (9.0N)k
(D) (5.0N)i – (1.0N)j – (1.0N)k
61. A circular disc is rotating about an axis perpendicular to the plane of the body and passing through the centre with angular velocity ‘ω1’. If the angular velocity of the disc is increased to ‘ω’, then moment of inertia of the disc :
(A) decreases
(B) increases
(C) remains same
(D) varies as a function of time
62. A small smooth spherical particle is placed on a smooth concave surface of radius R = 19.6 m. If it is displaced from the mean position, it executes SHM. The time period of oscillation will be _____.
(A) 2π
(B) 2√2π
(C) π
(D) √2π
63. If a wire is made ‘n’ times shorter than its original length (l), the effect on its elongation will be (a) ____ and the maximum load it can bear will be (b) _____.
(A) (a) no change (b) 1/n times the initial value.
(B) (a) 1/n times the initial value (b) no change
(C) (a) 1/n times (b) n times
(D) (a) n times (b) 1/n times
64. The V-I curve for a conductor is plotted at two different temperatures T1 and T2. If T1 > T2 then the slope of the V-I curve will be more at which temperature?
(A) Same of both temperatures
(B) T1
(C) Does not depend on temperature
(D) T2
65. A box of mass 5.0 kg is lying on a horizontal smooth surface. It is pulled by applying a force of 10.0 N at an angle of 30° to the horizontal. The magnitude of the acceleration of the box is about :
(A) 1.7 m/sec2
(B) 1.0 m/sec2
(C) 2.0 m/sec2
(D) 3.4 m/sec2
66. In 50 years, tritium undergoing beta decay reduced to one-sixteenth of its initial amount. The half-life of tritium is :
(A) 6.25 years
(B) 12.5 years
(C) 50 years
(D) 0.25 years
CHEMISTRY
Questions : 66
67. Which of the following elements or compounds does NOT dissolve in water ?
(A) Cane sugar
(B) Sulphur
(C) Urea
(D) Glucose
68. Which of the following molecule has bond order two?
(A) He2
(B) O2−1
(C) C2
(D) N2
69. What will be the ‘spin only’ magnetic moment of M3+ ion in aqueous solution for an element having Z = 26 ?
(A) 3.87 BM
(B) 5.92 BM
(C) 2.82 BM
(D) 4.89 BM
70. Which of the following devices is used for determining the resistance of a material?
(A) Wheatstone bridge
(B) Salt bridge
(C) Galvanometer
(D) Daniell cell
71. Select the option that is true regarding the following two statements labeled Assertion (A) and Reason (R)
Assertion (A) : Hoffmann’s ammonolysis of alkyl halides gives a mixture of primary, secondary, tertiary mines along with some quaternary ammonium halide.
Reason (R) : Primary amine with its lone pair of electron acts as a nucleophile.
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A
(C) A is false and R is true
(D) A is true but R is false
72. Which of the following is NOT a characteristic of an amorphous solid?
(A) They are isotropic
(B) They are anisotropic
(C) They do not have a pattern of arrangements
(D) They do not have a sharp melting point.
73. Select the correct statement from the following options.
(A) Colloids differ from crystalloids in electrical behavior of particles
(B) Colloids differ from crystalloids in solubility of the particles
(C) Colloids differ from crystalloids in nature of the particles.
(D) Colloids differ from crystalloids in size of the particles.
74. The functional group, which is NOT present in aspartame is :
(A) aldehydic group
(B) amino group
(C) ester group
(D) carboxylic group
75. What are the main components of phosphate rocks?
(A) Ca2(PO4)6CaF2
(B) Ca3(PO4)6CaF2
(C) Ca2(PO4)8CaF2
(D) Ca9(PO4)6CaF2
76. The ion having the largest size is :
(A) I−
(B) Br−
(C) Cl−
(D) F−
77. Which of the following statement is correct for oxides of halogens?
(A) I2O5 is a good oxidizing agent
(B) ClO2 is used as washing soda.
(C) BrO3 exist only at higher temperature
(D) Bromine oxides are less powerful oxidizing agent.
78. The oxidation of p-nitrotoluene with chromic trioxide in acetic anhydride followed by hydrolysis in aqueous acid gives :
(A) benzoic acid
(B) benzaldehyde
(C) p-nitrobenzaldehyde
(D) p-nitrobenzoic acid
79. Which of the following lanthanoids has an abnormally low value of third ionization enthalpy?
(A) Ytterbium
(B) Cerium
(C) Europium
(D) Lutetium
80. What temperature is required in the preparation of low-density polyethylene?
(A) 350-570 K
(B) 150-570 K
(C) 650-970 K
(D) 950-1270 K
81. Which of the following polymers is resistant to the action of petrol and lubrication oil?
(A) Melamine
(B) Nylon-6, 6
(C) Buna-N
(D) Butadiene-styrene polymer
82. What will be the hybrid state of BeCl2 in vapour state?
(A) sp3d
(B) sp
(C) sp3
(D) sp2
83. Which of the following crystals is an example of covalent crystal?
(A) Water
(B) Potassium Nitrate
(C) Carborundum
(D) Carbon dioxide
84. The composition of German silver is :
(A) Cobalt, zinc and silver
(B) Copper, zinc and tin
(C) Copper, zinc and silver
(D) Copper, zinc and nickel
85. Which of the following is the correct increasing order with respect to melting points?
(A) PH3 < AsH3 < SbH3 < NH3
(B) SbH3 < PH3 < AsH3 < NH3
(C) AsH3 < SbH3 < PH3 < NH3
(D) NH3 < PH3 < AsH3 < SbH3
86. Complete the balanced reaction.
Ca3P2 + 6H2O→
(A) 3CaO + 2PH3
(B) Ca(OH)2 + 2PH3
(C) 6CaO + 2PH3
(D) 3Ca(OH)2 + 2PH3
87. Which of the following coordination entities would have a square planar geometry?
(A) [Ni(H2O)4]2+
(B) [Ni(CO)4]
(C) [Ni(Cl)4]2−
(D) [Ni(CN)4]2−
88. Match the Column I (Structure) with Column II (IUPAC name) and select the correct answer using the codes given below.
(A) a-(iii), b-(i), c-(iii)
(B) a-(iii), b-(i), c-(ii)
(C) a-(i), b-(ii), c-(iii)
(D) a-(i), b-(ii), c-(iii)
89. Toulene on oxidation with alkaline potassium permanganate followed by hydrolysis gives compound X. X on reaction with ethanol in presence of conc.
H2SO4 gives Y. Compound X and Y respectively are :
(A) Benzoic acid; Ethyl-2-methyl benzoate
(B) 2-Methyl Benzoic acid; Ethyl benzoate
(C) 2-Methyl Benzoic acid; Ethyl-2-methyl benzoate
(D) Benzoic acid; Ethyl benzoate
90. Match the Column I with Column II and select the correct answer using the codes given below.
(A) (a)-I, (b)-II, (c)-III
(B) (a)-II, (b)-I, (c)-III
(C) (a)-I, (b)-III, (c)-II
(D) (a)-III, (b)-II, (c)-I
91. Match the Column I with Column II and select the correct answer using the codes given below:
(A) A-(II), B-(I), C-(III)
(B) A-(I), B-(III), C-(III)
(C) A-(III), B-(II), C-(I)
(D) A-(I), B-(II), C-(III)
92. What is the rate constant for the 1st order reaction?
(A) moll−1
(B) S−1
(C) mol L−1s−1
(D) mol−1LS
93. Which of the following dehydrating agents CANNOT convert ethanol into ethene?
(A) HCl
(B) H3PO4
(C) Conc. H2SO4
(D) D.Al2O3
94. Which of the following a consequence of Lanthanoid contraction?
(A) Atomic radius of Zn is larger than Ni and Cu.
(B) ∆a H increases from left to right in first transition series
(C) Zr and Hf have almost the same atomic radii
(D) Atomic radius decreases from left to right in second transition series.
95. Which of the following compounds has the lowest melting point?
(A) BaCl2
(B) NaF
(C) CaF2
(D) BeCl2
96. How many isomers are possible of dihalogen derivatives of propane?
(A) Four
(B) Three
(C) Five
(D) Two
97. Select the correct statement regarding Li.
(A) 1st ionization energy of Li is equal to Be.
(B) 1st ionization energy of Li is equal to F.
(C) 1st ionization energy of Li is lesser than Be.
(D) 1st ionization energy of Li is greater than to Be.
98. Which of the following polymers is NOT an example of synthetic polymer?
(A) Rayon
(B) Buna-S
(C) Polythene
(D) Nylon-6, 6
99. Which of the following is true about galvanic cell?
(A) It converts electrical energy into chemical energy
(B) It converts chemical energy into electrical energy.
(C) It converts mechanical energy into electrical energy.
(D) It converts electrical energy into mechanical energy.
100. How many O-O bonds present in the ozone molecule are identical?
(A) 0
(B) 2
(C) 1
(D) 3
101. Chloroform is NOT used as anaesthetic due to the formation of:
(A) chlorine gas
(B) carbon-dikoxide gas
(C) oxygen gas
(D) phosgene gas
102. What will be the change in entropy for the following process?
CaCO3(s) → CaO(s) + CO2(g)
(A) Entropy increases
(B) Entropy decreases
(C) Entropy becomes zero
(D) Entropy remains unchanged
103. Which of the following is correct?
(A) Neutrons = Atomic mass – Atomic number
(B) Neutrons = Atomic number – Atomic mass
(C) Protons = Atomic mass – Atomic number
(D) Electrons = Atomic mass/ atomic number
104. In a bomb calorimeter, energy change occurring in a reaction is measured under which of the following conditions?
(A) At constant temperature
(B) At constant pressure
(C) At constant pressure and volume
(D) At constant volume
105. Phenol on treatment with conc. H2SO4 at 373 K given compound A, which on treatment with excess of bromine water gives compound B. Compound A and B are respectively
(A) 4-phenolsulphonic acid; 4-Bromophenol
(B) 2-Phenolsulphoni acid; 2,4,6-Tribromophenol
(C) 2-Phenolsulphonic acid, 2-Bromophenol
(D) 4-Phenolsulphonic acid; 2,4,6-Tribromophenol
106. Which of the following statement is INCORRECT regarding the carbonyl group?
(A) The carbonyl carbon acts as nucleophile
(B) The carbonyl oxygen acts as a nucleophile
(C) The carbonyl carbon is sp2 hybridised
(D) It is polar in nature.
107. Which is the following statements is INCORRECT?
(A) Inductive effect is temporary effect while electrometric effect is permanent effect.
(B) Inductive effect involves the partial displacement of sigma electrons while in electrometric effect, complete transference of pi-electrons takes place.
(C) In inductive effect partial charges are developed on atoms and no ions are formed, while in electrometric effect complete transference of electrons takes place
(D) In this effect the displaced electron pair does not leave its orbitals but in electrometric effect it leaves its orbital.
(A) C
(B) B
(C) A
(D) D
108. What is the numerical value of Avogadro’s number?
(A) 6.022 × 1026
(B) 6.022 × 1023
(C) 6.022 × 1025
(D) 60.22 × 102
109. How many number of zero oxidation states of sulphur atoms are present in tetrathionate ion?
(A) 2
(B) 3
(C) 1
(D) 4
110. The slope of plot between ln [R] vs t give ____.
(A) Concentration of the products
(B) Molecularity of the reaction
(C) Rate of the reaction
(D) Rate constant
111. The compound BeCl2 is ____.
(A) Polymeric
(B) Ionic
(C) Co-ordinate
(D) Covalent
112. How many double bonds are present in butane?
(A) 1
(B) 0
(C) 2
(D) 3
113. The packing fraction of a simple cubic system is :
(A) 0.524
(B) 0.480
(C) 0.680
(D) 0.740
114. Choose the correct differential rate equation for the following reaction:
A + 3B → 2C + D
(A) r = −d[A]/dt = −1/3[B]dt = 1/2d[C]dt = d[D]/dt
(B) r = −d[A]/dt = 1/3d [B]/dt = 1/2d[C]/dt = d[D]/dt
(C) r = −d[A]/dt = 1/3d[B]/dt = −1/2d[C]dt = d[D]/dt
(D) r = −d[A]/dt = −3d[B]/dt = 1/2d[C]/dt = d[D]/dt
115. Which of the following correctly represents Gay Lussac’s law?
(A) V/T = K
(B) VT = K
(C) PT = K
(D) P/T = K
116. The number of primary –OH and secondary –OH group present in ethylene glycol is :
(A) One primary –OH and one secondary –OH group
(B) Only two secondary –OH groups
(C) Only two primary –OH groups
(D) Two primary –OH and one secondary –OH group
117. Which of the following statements is correct about bonding in metal carbonyls?
(A) The M-C sigma bond is formed by the donation of electron pair from a metal d-orbital into a vacant bonding orbital of a CO molecule.
(B) The M-C pi bond is formed by donation of electron pair from a metal d-orbital into vacant π* antibonding orbital of CO molecule.
(C) The M-C pi bond is formed by the donation of lone pair of electrons of carbonyl carbon into a vacant metal d-orbital
(D) The M-C sigma bond is formed by donation of electron pair from a metal d-orbital into vacant π* antibonding orbital of Co molecule.
118. Which of the following is NOT an antacid?
(A) Aluminium hydroxide
(B) Lansoprezole
(C) Ranitidine
(D) Chlorpheniramine
119. Match list I with list II and select the correct answer using the codes given below.
(A) a-ii, b-i, c-iv, d-iii
(B) a-ii, b-iii, c-i, d-iv
(C) a-iii, b-iv, c-ii, d-i
(D) a-iii, b-ii, c-iv, d-i
120. Select the elements can form d π – d π bond with transition metals.
(A) Arsenic, nitrogen and phosphorus
(B) Phosphorus and nitrogen
(C) Phosphorus and arsenic
(D) Arsenic and nitrogen
121. If the partial pressures of four non interacting gases are p1, p2, p3 and p4, then the pressure of mixture of these gases, P is _____.
(A) p1 + p2 + p3 + p4
(B) (p1 + p2) (p3 + p4)
(C) p1 × p2 × p3 × p4
(D)
122. Which of the following reagent is used in the preparation of K3[Fe(CN)6] from K4[Fe(CN)6]?
(A) O3
(B) MnO2
(C) H2O2
(D) AlH3
123. Which of the following is the increasing order of a s-block elements with respect to wavelength?
(A) Li < Na < K < Rb
(B) Na < Li < K < Rb
(C) Rb < K < Na < Li
(D) Na < K < Li < Rb
124. What is the nature of hexaflourides?
(A) Solid
(B) Gaseous
(C) Liquid
(D) Solid and liquid
125. Which of the following molecule will produce 2 moles of AgCl precipitate with an excess of AgNO3 solution?
(A) [Cr(NH3)5Cl]Cl2
(B) [Cr(NH3)3Cl3]
(C) [Cr(NH3)4Cl2]Br
(D) [Cr(NH3)6]Cl3
126. The shape and bond angle of 3° amine is :
(A) Pyramidal, 108°
(B) Tetrahedral, 108°
(C) Pyramidal, 107°
(D) Tetrahedral, 109.28°
127. The common name of (CH3)2C = CHCOCH3 is :
(A) isopropyl ketone
(B) di-isopropyl ketone
(C) acrolein
(D) mesityl oxide
128. Under which of the following conditions will a process be spontaneous at all temperature?
(A) ∆H = +ve, ∆S = +ve
(B) ∆H = −ve, ∆S = +ve
(C) ∆H = −ve, ∆S = −ve
(D) ∆H = +ve, ∆S = −ve
129. Select the correct pairs of isobars from the following.
130. Select the option that is true regarding the following two statements labeled Assertion (A) and Reason (R).
Assertion (A) : Tertiary alcohols are less acidic than secondary and primary alcohols.
Reason (R) : An electron releasing group increases the polarity of O-H bond by increasing the electron density on oxygen atom.
(A) Both A and R are true and R is the correct explanation of A.
(B) A is false and R is true
(C) Both A and R are true but R is not the correct explanation of A
(D) A is true but R is false
131. Which of the following is lyophobic in nature?
(A) Gelatin
(B) Sulphur
(C) Starch
(D) Protein
132. Which of the following is used as Tranquillizers?
(A) Iburofen
(B) Dichlorofenic sodium
(C) Veronal
(D) Naproxen
MATHEMATICS
Questions : 68
133. Which of the following is a statement?
(A) Oh ! we have drawn the match
(B) May God bless you !
(C) Jupiter is a planet
(D) I may live long !
134. The value of the integral is :
(A) −1
(B) ∞
(C) 1
(D) 0
135. Match the column A with column B
(A) 1 → P; 2 → P; 3 → S; 4 → R
(B) 1 → R; 2 → P; 3 → S; 4 → R
(C) 1 → Q; 2 → P; 3 → S; 4 → R
(D) 1 → Q; 2 → P; 3 → P; 4 → R
136. The value of
137.
(A) n – 1 + 2−n
(B) n – 1 – 2−n
(C) n + 1 – 2−n
(D) n + 1 + 2−n
138. In how many ways can 16 examination papers be arranged, so that the best and the worst papers never come together?
(A) 14 – 15!
(B) 15 – 14!
(C) 15 – 16!
(D) 16 – 15!
139. The value of
(A) 2
(B) 0
(C) −2
(D) 1
140. The number of elements in a set {x : x ϵ a all alphabets} is :
(A) 12
(B) 5
(C) 26
(D) 30
141. If f : R → A defined by f(x) = {x – 2} – 3 is a surjection then A is ____.
(A) (−∞, 1] ∪ [2, ∞)
(B) [−3, ∞)
(C) [1, 2]
(D) (1, ∞)
142. If ω is non-real cube root of unity, x = a + b, y = aω2 + bω and z = bω2 + aω, then x2 + y2 + z2.
(A) 12a2b2
(B) 12ab
(C) 6ab
(D) 6a2b2
143. For n ϵ N, 2.3n+1 + 3n + 36 is divisible by :
(A) 2
(B) 8
(C) 6
(D) 3
144. The polar form of −√7 + i√21 is :
145. Let then the value of tan(x/2) is :
(A) 15/29
(B) 14/29
(C) 13/25
(D) −15/29
146. The angle between the lines
x + y – 5 = 0, 7x + 5y – 8 = 0 is :
147. The product of the slopes of the tangents to the parabola y2 = x drawn from the point (1, −2) is :
(A) −1/4
(B) 1/4
(C) −2/4
(D) 2/4
148. The product of all the nth roots of unity is :
(A) 1
(B) 0
(C) −1
(D) (−1)n−1
149. The number an = 6n – 5n for n = 1, 2, 3,…. When divided by 25 leaves the remainder
(A) 9
(B) 7
(C) 3
(D) 1
150. The 11th term of sequence 3, 2, 4/3, 8/9….is :
(A) 210/69
(B) 910/39
(C) 210/39
(D) 210/310
151. The equation of a plane bisecting the angle between the planes 4x – 2y + 4z + 6 = 0 and 6x – 4y + 12z + 16 = 0 is :
(A) 23x + 13y + 32z – 45 = 0
(B) 5x – y – 4z – 3 = 0
(C) 23x – 13y + 32z + 5 = 0
(D) 5x – y – 4z – 45 = 0
152. If E is an impossible event, then P(E) is :
(A) 2
(B) 1
(C) 0
(D) 0.1
153. The equation of line through (1, 3) with slope 2 is :
(A) y = x + 2
(B) y = x – 2
(C) y = 2x – 1
(D) y = 2x + 1
154. Find the number of 2000th roots of unity which are 2020th roots of unity.
(A) 2
(B) 0
(C) 5
(D) 20
155. If 26Pr+6 : 25Pr+3 = 11050 : 1 then r is equal to :
(A) 16
(B) 4
(C) 6
(D) 8
156. Let S be the set of people living in India, then the relation R in S defined by ‘aRb if a and b have same father’ is :
(A) reflexive
(B) equivalence
(C) symmetric
(D) transitive
157. If and
then the value of ∝ for which A2 = B is :
(A) 2
(B) −1
(C) 1
(D) 0
158. Match the columns
(A) 1 → Q, 2 → Q, 3 → R, 4 → P
(B) 1 → S, 2 → Q, 3 → R, 4 → P
(C) 1 → S, 2 → Q, 3 → P, 4 → P
(D) 1 → S, 2 → R, 3 → R, 4 → P
159. The solution of the differential equation is :
160. If & B = adj A & C = 5A, then the value of
is _____.
(A) 1
(B) 2
(C) 0
(D) −1
161. The exponent of 6 in 100! is :
(A) 45
(B) 48
(C) 13
(D) 22
162. In a city 15% are suffering from fever and 10% are suffering from cold and 5% are suffering from both. The probability that they are suffering from either cold or fever is :
(A) 50%
(B) 30%
(C) 20%
(D) 40%
163. If A = {−3, 1}, B = {x ∈ R : x2 + 2x – 3 = 0}, and C = {1, 2, 3} and D = {x ∈ R : x2 – 6x2 + 11x – 6 = 0}, then the equal sets are :
(A) A, B and C
(B) A and B
(C) A and C
(D) A, C and D
164. If such that |A – KI| = 0, then the sum of all the possible values of K is ____.
(A) 0
(B) 2
(C) 6
(D) 4
165. If nA = m and nB = 2, then the number of possible surjective mappings from A to B is :
(A) m!
(B) 2m
(C) 2m – 1
(D) 2m – 2
166. A line segment has length 1089 and direction ratios are 6, −2, 9. If the line makes an obtuse angle with x-axis, the components of the line vector are :
(A) −594, 198, 891
(B) 27, −18, 54
(C) −27, 18, −54
(D) 18, −6, 27
167. The set {x : x is a positive interger less than 5 and 5x – 1 is an even number} in roster form is :
(A) {2, 4, 6}
(B) {1, 3, 5}
(C) {1, 2, 3, 4, 5, 6}
(D) {1, 2, 3, 4}
168. The solution of the differential equation x(x + y) dy – y2dx = 0 is
(A) y = −x log cy
(B) y = x log cxy
(C) y = x log cy
(D) y = −x log cxy
169. The contrapositive of the statement, ‘IF I am not happy, then I will go to the picture’ is :
(A) I I am happy, then I will not go to the picture
(B) If I will go to the picture, then I am happy
(C) If I will go to the picture, then I am not happy
(D) I I will not go to the picture, then I am happy
170. What is the probability of getting a sum as 7 or 8 with two dice ?
(A) 1/36
(B) 1/12
(C) 5/36
(D) 11/36
171. If the 4th and 9th term of a GP be 54 and 13122, respectively, then its second term is :
(A) 10
(B) 12
(C) 6
(D) 8
172. If f(x) = ex, g(x) = tan−1 x and k(x) = f(g(x)) then
(A) 1 – x2
(B)
(C)
(D) 1 + x2
173. If x = sin−1 t, y = log(1 – t2), then is _____.
(A) −√3/2
(B) −2/√3
(C) 2/√3
(D) √3/2
174. If the eccentricity of the hyperbola is √3 then the eccentricity of its conjugate hyperbola is :
(A) 1/2
(B)
(C)
(D) 3/2
175. The eccentricity and length of latus rectum of hyperbola
X2 – 2y2 – 6x + 8y = 36 is :
(A) √3 and √14
(B) √13 and √37
(C)
(D) 2 and √3
176. If A and B are matrices of order 3 × 3 and if |A| − 2, |B| = 3, then |4A2B3| is ____.
(A) 7062
(B) 7612
(C) 6912
(D) 6072
177. The solution of the differential equation is :
178. Find the equation of the line through the intersection of x + y – 1 = 0, 2x – y – 2 = 0 and perpendicular to 2x – 3y + 2 = 0 is :
(A) 3x + 2y – 3 = 0
(B) 3x + 2y + 3 = 0
(C) 3x – 2y – 3 = 0
(D) 3x + 2y – 2 = 0
179. If R is relation on the set A = {1, 2, 3}, then R = {(1, 1), (2, 2), (3, 3)} is a :
(A) Transitive
(B) Reflexive
(C) All the given options
(D) Symmetric
180. The value of
(A) 100√2
(B) 50 π
(C) 100 π
(D) 50√2
181. Let m = tan2(sec−12) + cot2(cosec−12), then the value of m3 – m is :
(A) 121
(B) 210
(C) 144
(D) 81
182. Match the following :
(A) A-i, B—ii, C-iii, D-iv
(B) A-ii, B-iii, C-iv, D-i
(C) A-iii, B-i, C-iv, D-ii
(D) A-iv, B-iii, C-ii, D-i
Note : For this question, discrepancy is found in question/answer. Full marks is being awarded to all candidates.
183.
184. If PA. = 0.25 PB. = 0.5, P(A ∪ B) = 0.59, then is :
(A) 1
(B) 0.09
(C) 0.9
(D) 0.2
185. The derivative of y = x3x sin(x) is :
(A) x3xsin(x)(3 + 3 ln(x) + cot (x))
(B) xx sin(x)
(C) x3x sin(x) cot (x)
(D) 3 + 3ln (x) + cot (x)
186. The value of is :
(A)
(B)
(C) loge 2
(D) 0
187. If then
(A) 0
(B) 2
(C) 1
(D) −1
188. If y = (loge x)x, then
189. There are 200 students in a class. In the examination, 100 of them failed in Mathematics, 90 failed in Physics, 80 failed in Biology and 64 failed in exactly two of the three subjects. Only two students passed in all the subjects. Then, the number of students failing in all the three subjects is :
(A) 2
(B) 6
(C) 4
(D) 8
190. Consider the following statements :
P : Pranav is skilled
Q : Pranav is poor
R : Pranav is honest
The negation of the “Pranav is Skilled and dishonest if and only if Pranav is poor” can be expressed as :
(A) ~(P ^ ~R) ↔ Q
(B) ~ (Q ↔ (P ^ ~ R))
(C) ~ p ^ (Q ↔ ~ R)
(D) ~ Q ↔ ~ P ^ R
191. The remainder obtained when
(n2 + 5n + 4) . (n2 + 5n + 6) + 31 is divided by 24 is :
(A) 9
(B) 7
(C) 25
(D) 32
192. If the function f : R → R defined by find f(x) + (1 – x)
(A) 0
(B) 2
(C) 1
(D) 22
193. The centre and radius of the circle x2 + y2 – 4x – 4y – 1 = 0 is :
(A) (1, 1) and 3
(B) (1, −2) and 3
(C) (2, −2) and 2
(D) (2, 2) and 3
194. If the first three terms in AP are 3, 7, 11 then the sum of the first 10 terms is :
(A) 222
(B) 211
(C) 223
(D) 210
195. The sequence of numbers are in :
(A) GP
(B) None of these
(C) HP
(D) AP
196. Match the column A with column B based on permutation and combination
| Column A | Column B |
| 1. There are 6 candidates for 3 posts. In how many ways can the posts be filled. | P. 180 Ways |
| 2. From among the 36 teachers in a school one principal and one vice-principal and one vice-principal are to be appointed. In how many ways can this be done. | Q. 1260 ways |
| 3. There are 15 buses running between Delhi and Mumbai. In how many ways can a man go to Mumbai and return by a different bus | R. 120 ways |
| 4. A teacher of a class wants to set on question from each of the two exercise in a book. If there are 15 and 12 questions in the two exercises respectively, then in how many ways can the two questions be selected. | S. 210 ways |
(A) 1 → Q; 2 → R; 3 → S; 4 → P
(B) 1 → R; 2 → Q; 3 → S; 4 → P
(C) 1 → R; 2 → R; 3 → S; 4 → P
(D) 1 → R; 2 → Q; 3 → P; 4 → P
197. In a college tournament, in which the participants were to play one game with another, two class players did not participate, after having played 2 games each. If the total number of games played is 82, then the number of participants at the beginning, was :
(A) 15
(B) 22
(C) 25
(D) 10
198. The equation of line which makes the intercepts 1 and 2 on x and y axis is :
(A) 3x – y – 2 = 0
(B) 3x + y – 2 = 0
(C) 2x – y – 2 = 0
(D) 2x + y – 2 = 0
199. Let f be the subset of R × R defined by f = {(x, y) + x2 + y2 = 9}. Here, f is :
(A) one-one
(B) onto
(C) none of these
(D) bijective
200. Cosine of the angle between the lines whose vector equations are
R = 5i + 3j – 6k + λ)(2i + j + 2k) and
r = 7i – 6k + μ(i + 2j + 8k);
(λ, μ being parameters,) is :
(A) 23/3√69
(B) 23/√69
(C) 20/3√69
(D) 20/√69
BIOLOGY
Question : 68
133. Animals in which sexes are separate, that is, males and females are distinct are known as :
(A) dioecious
(B) hermaphrodite
(C) transgender
(D) monoecious
134. Identify the INCORRECT statement.
(A) For bacteria to take up plasmid, the cells must be rendered DNA – competent.
(B) Microparticles of platinum are utilized in gene gun.
(C) Biolistics allows for the direct injection of recombinant DNA.
(D) Disarmed pathogen vectors deliver recombinant DNA to hosts.
135. Which RNA acts as genetic material?
(A) rRNA
(B) mRNA
(C) tRNA
(D) TMV RNA
136. Match the items given in column I with their functions in column II.
(A) a-ii, b-iii, c-iv, d-i
(B) a-iii, b-ii, c-i, d-iv
(C) a-iii, b-iv, c-ii, d-i
(D) a-iii, b-ii, c-iv, d-i
137. Which chlorophyll type is a primary, universal photosynthetic pigment and acts as reaction centre?
(A) Chlorophyll d
(B) Chlorophyll c
(C) Chlorophyll b
(D) Chlorophyll a
138. Arrange the following events of an action potential in the proper sequence.
(a) Repolarisation (b) Influx of Na+
(c) Efflux of Na+ (d) Depolarisation
(A) c, d, b, a
(B) b, a, c, d
(C) b, d, c, a
(D) b, c, d, a
139. Which genetic disorder is sex-linked (X chromosome) and shows effect in the defective form of blood clotting factor VIII/IX ?
(A) Sikle-cell anaemia
(B) Haemophilia A/B
(C) Phenylketonuria
(D) Huntington’s disease
140. Rod-like structure, called notochord, is not present in animals belonging to Porifera to Echinodermata, they are called :
(A) chordates
(B) non-chordates
(C) fishes
(D) vertebrates
141. How many ATP are required for the synthesis of one glucose molecule in C3 pathway?
(A) 12 ATP
(B) 38 ATP
(C) 12 ATP
(D) 30 ATP
142. The matrix of mitochondria has :
(A) Single circular DNA
(B) Double stranded straight DNA
(C) Double stranded circular DNA
(D) Single straight DNA
143. What is the shape of a simple stirred-tank bioreactor?
(A) A rectangular tank
(B) Round bottom flask
(C) Cylindrical tank
(D) Volumetric flask
144. In prokaryotic cells mesosomes do not help in :
(A) Hormone formation
(B) Respiration
(C) Cell wall formation
(D) DNA replication
145. Which of the following symptoms is true about elephantiasis?
(A) Obstructed intestinal passage
(B) Respiratory problems
(C) Genital organs are also frequently affected
(D) Various parts of the body are covered in scaly lesions
146. Eggs and sperms are produced by the same individual are called :
(A) heterosexual
(B) intersex
(C) dioecious
(D) hermaphrodite
147. Mitochondria divides by:
(A) fusion
(B) budding
(C) fission
(D) vegetative propagation
148. In which of the following organisms – blood pumped by the heart is always circulated through a closed network of blood vessels?
(A) Periplanata and Balanoglossus
(B) Ascaris and Taenia
(C) Pheretima and Rana
(D) Rana and Balanoglossus
149. Sponges have a water canal system, in which water enters through ostia and goes out through :
(A) coelom
(B) pseudocoelom
(C) osculum
(D) coelenteron
150. The property of contact inhibition of cells is lost in ______ patients.
(A) HIV
(B) allergy
(C) cancer
(D) diabetes
151. Select the correctly matched set from the following options.
(A) Birth canal : Uterus, cervical canal, vagina
(B) Tertiary follicle : Antrun, theca layers, secondary oocyte
(C) Female accessory ducts : Ovulation, fertilization, implanation.
(D) Female external genitalia : Vagina, mons pubis, hymen, clitoris
152. Which syndrome arises due to translocation of the major portion of chromosome 21 to chromosome 14?
(A) Klinefelter syndrome
(B) Turner syndrome
(C) Down’s syndrome
(D) Phenylketonuria
153. The gaps between two adjacent myelin sheaths of a myelinated nerve fibre is called :
(A) chemical synapse
(B) synaptic cleft
(C) nodes of Ranvier
(D) electrical synapse
154. Select the INCORRECT combination.
(A) Free nuclear endosperm – Ground nut
(B) Cellular endosperm – Datura
(C) Non endospermic seed – Bean
(D) Endospermic seed – Castor
155. The prokaryotic cells are NOT represented by :
(A) mycoplasma
(B) blue-green algae
(C) amoeba
(D) bacteria
156. During reflex action, signal from a sensory organ transmits the impulse via a _____ into the spinal cord (CNS).
(A) ventral nerve root
(B) inter neuron
(C) efferent pathway
(D) dorsal nerve root
157. Chondrichthyes and Osteichthyes are classes in which fins are present for locomotion. They belong to the group :
(A) Reptilia
(B) Tetrapoda
(C) Amphibia
(D) Pisces
158. Which of the following statements regarding human reproduction are INCORRECT ?
(a) The placenta develops within the ovarian cavity.
(b) The placenta is present between the umbilical cord and the embryo.
(c) Relaxin is an exclusive secretion of the placenta
(d) Placenta is formed due to inter digitation of chorionic villi and myometrial tissue.
(A) a, b and c
(B) a, b, c and d
(C) b and d
(D) b, c and d
159. Identify the invertebrate
(A) Fishes
(B) Insects
(C) Amphibians
(D) Reptiles
160. Which of the following is NOT correctly matched?
(A) Lysine – basic amino acid
(B) Uridylic acid – nucleotides
(C) Valine – neutral amino acid
(D) Glycerol – disaccharide
161. Osmolality of the glomerular filtrate can be 1200m Osmoi L−1 in which of the following part
(A) DCT in cortex
(B) Bowman’s capsule in outer medulla
(C) Hair pin bend of Henle’s loop in inner medulla
(D) PCT in contex
162. Which part of the pituitary is under the direct neural regulation of the hypothalamus?
(A) Pars distalis
(B) Pars intermedia
(C) Neurophypophysis
(D) Adenohypophysis
163. Electron hole is formed in which chlorophyll reaction centre of photosynthesis?
(A) Chlorophyll a675
(B) Chlorophyll a683 (P680)
(C) Chlorophyll a673
(D) Chlorophyll a703(P700)
164. In leucoplasts of plant cells, elaioplasts store :
(A) Carbohydrates
(B) Oils and fats
(C) Starch
(D) Proteins
165. In which of the following places can antigens bind?
(A) Place : 1 and 2
(B) Place : 1, 2 and 3
(C) Place : 1 and 3
(D) Place : 2 and 3
166. Organisms having dorso-ventrally flattened body and known as ‘flatworms’ are classified under the phylum:
(A) Annelida
(B) Mollusca
(C) Platyhelminthes
(D) Porifera
167. Match the terms in column I with their descriptions in column II.
(A) a-iv, b-ii, c-i, d-iii
(B) a-iii, b-iv, c-ii, d-i
(C) a-ii, b-i, c-iii, d-iv
(D) a-iii, b-i, c-ii, d-iv
168. Mendel proposed four postulates, where postulate 4 is referred to as the :
(A) purity of gametes
(B) principal of independent assortment of factors
(C) dominant unit factor
(D) principal of segregation
169. Which of the following bacteria is capable of producing DNA polymerase?
(A) Thermus aquaticus
(B) Escherichia coli
(C) Bacillus thurengensis
(D) Agrobacterium tumifaciens
170. Sella turcica is the depression in the sphenoid bond which lodges:
(A) pituitary gland
(B) thyroid
(C) pineal gland
(D) thymus
171. Second messengers generates response to all EXCEPT :
(A) Cortisol
(B) Thyrocalcitonin
(C) Parathyroid hormone
(D) Erythropoietin
172. Jaintia Hills in Meghalaya is an example of ______.
(A) biosphere reserves
(B) wildlife safari parks
(C) zoological parks
(D) sacred groves
173. What is the major RBC producing organ in our body ?
(A) Tonsils
(B) Bone marrow
(C) Thymus
(D) Spleen
174. Which genetic disorder results in mental retardation and is inherited as an autosomal recessive trait?
(A) Phenylketonuria
(B) Sickle-cell anaemia
(C) Klinefelter syndrome
(D) Down’s syndrome
175. The primary acceptor molecule Ribulose1, 5-biphosphate (RuBP) of Co2 fixation cycle was found by which scientist?
(A) Basham
(B) CR Slack
(C) Dicker
(D) MD Hatch
176. In conditions of low intensity of light and increases in temperature, the rate of photosynthesis will :
(A) remain constant
(B) decrease
(C) rapidly increase
(D) increase
177. Lysosomal vesicles do NOT contain :
(A) Proteases
(B) insulin
(C) carbohydrates
(D) lipases
178. In which place in 1856, Mendel started his experiments on controlled hybridization in garden peas?
(A) Monstery garden at Heinzen Dorf
(B) Monsastery garden of Bruno
(C) Monastery garden at Hopkins University
(D) Monastery garden at Vienna
179. When breathing causes wheezing it is a symptom of which of the following disease?
(A) Emphysema
(B) Gout
(C) Jaundice
(D) Asthma
180. In the context of reproductive health, ART refers to :
(A) artificial method and techniques to prevent pregnancy.
(B) intentional or voluntary termination of pregnancy before full term.
(C) special techniques which assist infertile couples to have children
(D) methods for the prevention and treatment of STIs.
181. Which of the following hormonal disorders is associated with the formation of harmful compounds known as ketone bodies?
(A) Diabetes insipidus
(B) Cretinism
(C) Diabetes mellitus
(D) Dwarfism
182. Which cell organelle is ABSENT in almost all plant cells ?
(A) Centrioles
(B) Ribosomes
(C) Plastids
(D) Mitochondria
183. The ultimate goal of practically all recombinant technologies is to generate a desirable_____.
(A) DNA
(B) bacteria
(C) vectors
(D) protein
184. Then +1 gamete is formed by the failure of paired homologus to separate at anaphase, process known as non-disjunction, appeared in :
(A) Phenylketonuria
(B) Down’s syndrome
(C) Sickle-cells anaemia
(D) Turner syndrome
185. Identify the INCORRECT statement about adaptation :
(A) Many adaptations are genetically predetermined.
(B) Some desert plants, such as Opuntia, do not have leaves.
(C) Leaf surfaces of many desert plants are covered in a thick layer of cuticle.
(D) Penguin may concentrate their urine to require less water to excrete
186. ______ are areas allocated for wildlife where they can freely utilize their habitats and natural resources.
(A) Wildlife Sanctuaries
(B) National parks
(C) Protected forests
(D) Biosphere Reserves
187. Chitinase can be found in which organism?
(A) Bacteria
(B) Virus
(C) Fungi
(D) Protozoa
188. During photosynthesis, assimilatory power ATP only synthesizes in which process of photosynthesis?
(A) cyclic photophosphorylation
(B) Non-cyclic photophosphorylation
(C) Photolysis
(D) Photosystem-II
189. Identify the true statement related to immunity.
(A) There are eight peptide chains in each antibody molecule
(B) A non-specific form of defence is acquired immunity
(C) Interferons are proteins secreted by virus-infected cells.
(D) T-lymphocytes are responsible for the production of antibodies.
190. Which of the following is NOT a part of innate immunity?
(A) Cellular barriers
(B) Blood-brain barriers
(C) Physical barriers
(D) Cytokine barriers
191. GIFT is an assisted reproductive technology indicated for a woman :
(A) Who wants to get rid of unwanted pregnancy
(B) Who cannot provide a suitable environment for fertilization
(C) Whose uterus does not support implantation
(D) Who cannot produce an ovum
192. Which of the following is NOT an altitude sickness symptom ?
(A) Heart palpitations
(B) Nausea
(C) Fatigue
(D) Anaemia
193. In an aquatic plant like water lily pollination is brought about by :
(A) Water
(B) Insects only
(C) Wind or insects
(D) Wind only
194. The characteristic feature of some animals, in which body is divided externally and internally into segments with serial repetition of some organs, is called :
(A) coelenteron
(B) spongocoel
(C) mesoderm
(D) metamerism
195. In which phase of the uterine cycle does implantation occurs ?
(A) Menstrual phase
(B) Proliferative phase
(C) Ovulatory phase
(D) Secretory phase
196. Which plants can tolerate high temperature and have greater productivity of biomass?
(A) C3 plants
(B) CAM plants
(C) C2 plants
(D) C4 plants
197. Which cycle plants are adapted to overcome photorespiration and deliver Co2 directly to the enzyme RuBisCo ?
(A) CAM cycle plants
(B) C2 cycle plants
(C) C4 cycle plants
(D) C3 cycle plants
198. How many hormones given below are increased several fold in the maternal blood during pregnancy?
(a) Thyroxine (b) Progesterone
(c) Cortisol (d) Oestrogens
(e) Prolactin
(A) 5
(B) 3
(C) 4
(D) 2
199. Which genetic disorder is inherited as a dominant trait?
(A) Cystic fibrosis
(B) Colour blindness
(C) Huntington’s disease
(D) Phenylketonuria
- The structure similar to notochord that is present in the collar region of Balanoglossus is called :
(A) cartilage
(B) vertebrae
(C) stomochord
(D) nerve chord
























 satisfied A2 + aA + bI = 0, then a, b are respectively equal to
satisfied A2 + aA + bI = 0, then a, b are respectively equal to then
then  is
is
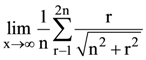 equals
equals decreases for all x, then
decreases for all x, then then at x = 0
then at x = 0
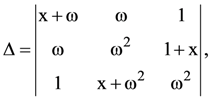 then value of x is
then value of x is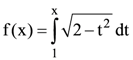 Then the real roots of the equation x2 – f′(x) = 0 are
Then the real roots of the equation x2 – f′(x) = 0 are is ([x] is the greatest integer function)
is ([x] is the greatest integer function)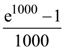
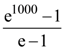
 then x is any term of the following.
then x is any term of the following.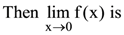
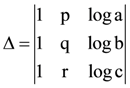
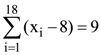 and
and  then the standard deviation of x1, x2, ….., x18 is
then the standard deviation of x1, x2, ….., x18 is
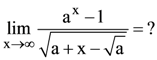
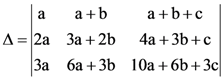






















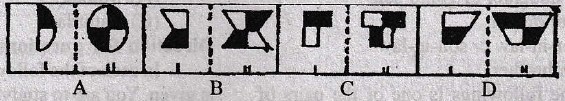






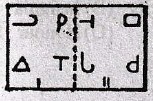



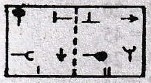








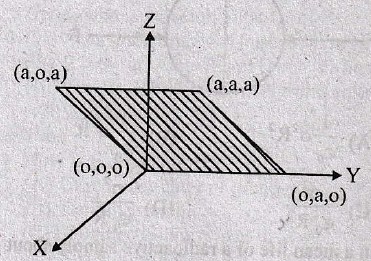
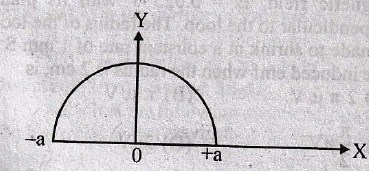
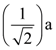

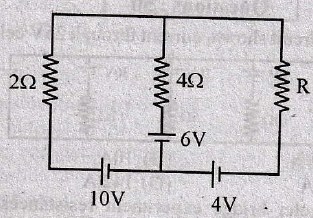







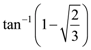


 will be
will be
 is incident on a horizontal glass-air surface at the critical angle. If a thin layer of water
is incident on a horizontal glass-air surface at the critical angle. If a thin layer of water  is poured on the glass-air surface. The angle at which the ray emerges into air at the water-air surface is
is poured on the glass-air surface. The angle at which the ray emerges into air at the water-air surface is


 The x coordinate of the particle at time
The x coordinate of the particle at time  would be
would be

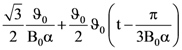

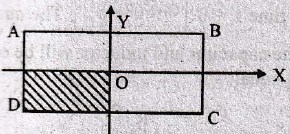








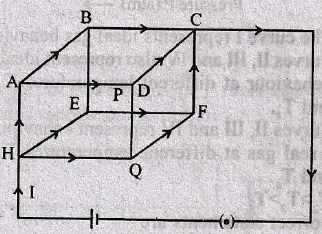
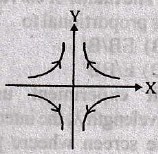



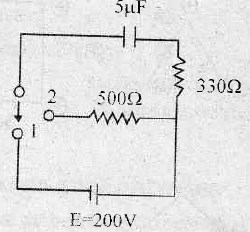
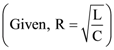 The current through the capacitor and inductor will be equal at a time t equals to
The current through the capacitor and inductor will be equal at a time t equals to








 is
is are equal, then value of n is
are equal, then value of n is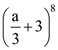 is 1120, then value of a is
is 1120, then value of a is is
is
 is equal to
is equal to

 are (respectively)
are (respectively) then
then then numerical value of ∆ is
then numerical value of ∆ is



 then A2 – 5A – 1 equals
then A2 – 5A – 1 equals is
is is
is





















 holds is
holds is







 is invetible for
is invetible for then k is
then k is is continuous at x = 0, is
is continuous at x = 0, is at the point (a cos θ, b sin θ) is
at the point (a cos θ, b sin θ) is then A is equal to
then A is equal to are
are then the value of m is
then the value of m is is
is is
is is
is is
is is
is

















 is
is
 are
are then the value of n is
then the value of n is is
is then
then is
is















 are
are
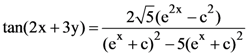

 is
is



 is
is is
is






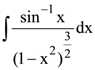 is given by
is given by



 then the values of x, y and z are respectively
then the values of x, y and z are respectively Then [AB]−1 is equal to
Then [AB]−1 is equal to



 is
is




 then x = ?
then x = ?



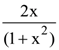
 then
then  then p = ?
then p = ?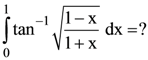



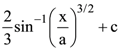



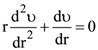




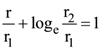 and r2 = er, then
and r2 = er, then 
 then the locus of P is
then the locus of P is




 is to be continuous at x = 0, then f(0) = ?
is to be continuous at x = 0, then f(0) = ? is discontinuous at x = ?
is discontinuous at x = ? is
is is
is
 is
is is
is
 is equal to
is equal to
 is
is is
is




 is
is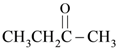
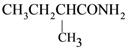 is
is
 is
is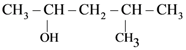 is
is







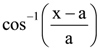
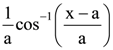
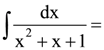
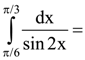
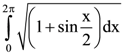 is
is is
is
 is equal to
is equal to
 whose distances from the centre of the ellipse is 2, is
whose distances from the centre of the ellipse is 2, is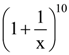 is
is is
is is
is

 where α, β & γ are the angles made by a line with the positive directions of the axes of reference, then the measure of θ is
where α, β & γ are the angles made by a line with the positive directions of the axes of reference, then the measure of θ is
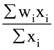
















 is equal to
is equal to
 is equal to
is equal to is equal to
is equal to is equal to
is equal to then f ‘(x) is equal to
then f ‘(x) is equal to


 then the value of a is
then the value of a is is
is then for every positive integer n, An is equal to
then for every positive integer n, An is equal to


 is nilpotent of index
is nilpotent of index and f(x) = 2x2 – 4x + 5, then f(A) is equal to
and f(x) = 2x2 – 4x + 5, then f(A) is equal to
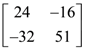


 is
is and (aI2 + bA)2 = A, then
and (aI2 + bA)2 = A, then is
is is
is is
is is
is









































 is an identity matrix.
is an identity matrix.





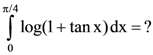
 is equal to
is equal to is
is


















 then det(A−1) = ?
then det(A−1) = ? then Fʹ(x) = ?
then Fʹ(x) = ?


 is
is
 then f(A) = ?
then f(A) = ?


 and
and  Then the value of F is
Then the value of F is







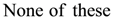


 is
is








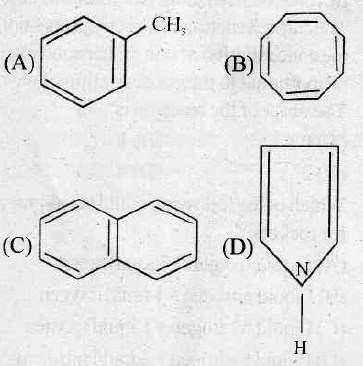
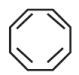
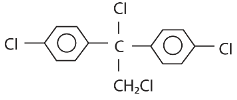
 is aromatic because it has
is aromatic because it has
 is
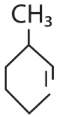
is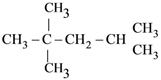






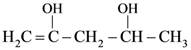
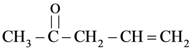




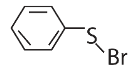


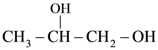
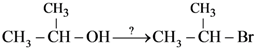
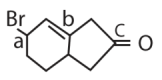
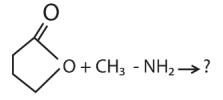
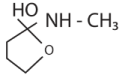


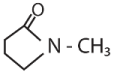

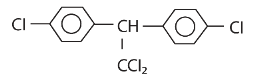

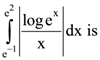
 |W| = 1, then Z lies on
|W| = 1, then Z lies on has the value
has the value has the value
has the value is
is is
is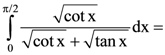
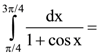
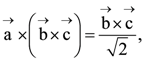 then the angle between
then the angle between 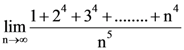
 has local minimum at x = ?
has local minimum at x = ?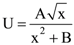

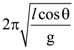

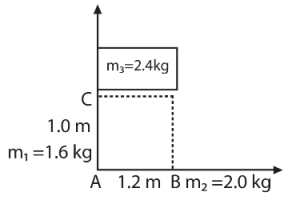
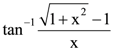 w.r.t.
w.r.t.  where p and q are integers, is equal to
where p and q are integers, is equal to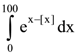 is ([ ] denotes the greatest integer function)
is ([ ] denotes the greatest integer function) and the plane 2x – 2y + z = 5 is
and the plane 2x – 2y + z = 5 is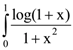
 is
is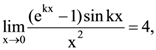 then k = 0
then k = 0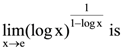
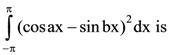
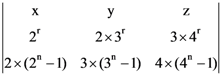 then
then