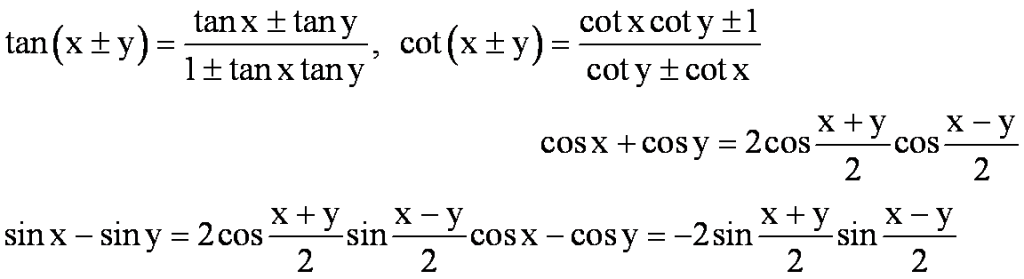AIEEA Conduct of Exam
AIEEA UG 2015 Scheme of Examination
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Date of Examination:
11th April, 2015
Duration:
2½ hours, Time 10.00 A.M. to 12:30 P.M.
Subject Streams, Question Paper and Writing Answers in the Examination
(i) The examination shall be conducted in one of the two subject streams namely ‘Stream-A’ comprising of Physics, Chemistry and Agriculture/Biology subjects and ‘STREAM-B’ comprising of Physics, Chemistry and Mathematics subjects.
(ii) Questions for Physics and Chemistry subjects shall be common for both subject streams.
(iii) Paper for ‘Stream–A’ shall carry questions on Biology as well as Agriculture subject, out of which the candidate has to attempt/opt/choose only one subject, either Biology or Agriculture. ‘Stream–B’ paper carries questions on Mathematics subject.
(iv) Question paper will have 180 multiplechoice objective type questions, each with four options, 60 questions in Physics, serially numbered from 1-60, 60 questions
in Chemistry, numbering from 61-120 and 60 questions in Agriculture/Biology/Mathematics, numbering from 121-180.
(v) Marking Scheme: Each correctly answered question will earn four marks, with a maximum of 720 marks for the paper. For each incorrect answer one mark will be deducted from the total score. Question with no response indicated will not be awarded any mark and there will be no negative marking for that question. The candidates are advised not to attempt such questions in the OMR answer sheet, for which they are not sure of the correct answer. More than one answer indicated against a question will be deemed as incorrect answer and will invite negative marking.
(vi) Candidate will be required to choose the correct answer and mark in the OMR answer sheet by darkening the corresponding circle/ bubble against the serial number of the question with black/ blue ball-point pen.
(vii) Question paper for the examination shall be bilingual, i.e., in ENGLISH and HINDI. In case of any variation in English and Hindi versions of question(s), English version will be taken as the final and correct version.
(ix) A visually challenged (blind) candidate, can request for the services of amanuensis (scribe) who must be one grade junior in academic qualification i.e not more than X/Matriculation pass in the present case and should not be related to the candidate. For amanuensis, the candidate must submit a separate request letter, at least one week in advance, to the concerned Nodal Officer of the Examination City Centre giving a copy of the PD/PC certificate issued by a competent authority. The services of amanuensis (Scribe) who should be from academic discipline other than that of the candidate, shall be provided free of cost by the Noda Officer. The scribe will have to give a suitable undertaking,confirming that the scribe fulfills all the stipulated eligibility criteria for a scribe as mentioned above. In case later on it is proved that candidate has suppressed material facts, the candidature of the applicant will stand cancelled, irrespective of the outcome of result of the written test. Such candidates who use a scribe shall be eligible for extra time of 50 minutes during examination.
AIEEA UG 2015 Updates
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
ICAR AIEEA UG 2015 information will be updated very shortly…
- ICAR National Talent Scholarship (NTS)
- AIEEA – UG – 2015 Introduction
- AIEEA – UG – 2015 Admit Card
- AIEEA – UG – 2015 Online Application
- AIEEA – UG – 2015 Conduct of Examination
- AIEEA UG Degree Programmes (ICAR)
AIEEA UG 2015 Exam Pattern
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
- The examination shall be conducted in one of the two subject streams namely
- ‘Stream-A’ comprising of Physics, Chemistry and Agriculture/Biology subjects.
- ‘Stream-B’ comprising of Physics,Chemistry and Mathematics subjects.
- Questions for Physics and Chemistry subjects shall be common for both subject streams.
- Paper for ‘Stream–A’ shall carry questions on Biology as well as Agriculture subject, out of which the candidate has to attempt/opt/choose only one subject, either Biology or Agriculture. ‘Stream–B’ paper carries questions on Mathematics subject.
- AIEEA Question Paper will have 180 Multiple Choice type questions, each with four options.
- 60 questions in Physics, serially numbered from 1-60,
- 60 questions in Chemistry, numbering from 61-120
- 60 questions in Agriculture/Biology/Mathematics, numbering from 121-180.
- Each correctly answered question will earn four marks, with a maximum of 720 marks for the paper.
- The duration of the exam would be 2½ hours.
- The Entrance Examination
- For each incorrect answer one mark will be deducted from the total score.
- Question with no response indicated will not be awarded any mark and there will be no negative marking for that question.
- The candidates are advised not to attempt such questions in the OMR answer sheet, for which they are not sure of the correct answer.
- More than one answer indicated against a question will be deemed as incorrect answer and will invite negative marking.
- Candidate will be required to choose the correct answer and mark in the OMR answer sheet by darkening the corresponding circle/ bubble against the serial number of the question with black/ blue ball-point pen.
-
Question paper for the examination shall be bilingual, i.e., in ENGLISH and HINDI. In case of any variation in English and Hindi versions of question(s), English version will be taken as the final and correct version.
-
A visually challenged (blind) candidate, can request for the services of amanuensis (scribe) who must be one grade junior in academic qualification i.e. not more than X/ Matriculation pass in the present case and should not be related to the candidate. For amanuensis, the candidate must submit a separate request letter, at least one week in advance, to the concerned Nodal officer of the Examination City Centre giving a copy of the PD/PC certificate issued by a competent authority. The services of amanuensis (Scribe) who should be from academic discipline other than that of the candidate, shall be provided free of cost by the Nodal Officer.
- The scribe will have to give a suitable undertaking, confirming that the scribe fulfills all the stipulated eligibility criteria for a scribe as mentioned above. In case later on it is proved that candidate has suppressed material facts, the candidature of the applicant will stand cancelled, irrespective of the outcome of result of the written test. Such candidates who use a scribe shall be eligible for extra time of 50 minutes during examination.
AIEEA UG 2015 Exam Centres
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
There are 49 exam centres allotted for ICAR – AIEEA 2015 examination. Given below the list of exam centres with their contact telephone numbers.
| AIEEA Examination Centres | ||
| Examination Center (Code No.) |
Name | Contact Phone No. of |
| 01 | Anand | 02692-271602 |
| 02 | Bengaluru | 080-28466353 |
| 04 | Bhubaneswar | 0674-2300060 |
| 06 | Bikaner | 0151-2250147 |
| 07 | Chennai | 044-24610565 |
| 08 | Cochin | 0484-2394867 |
| 09 | Coimbatore | 0442-2472986 |
| 10 | Delhi | 011-25843382 |
| 11 | Dharwad | 0836-2792180 |
| 12 | Gangtok | 03592-231030 |
| 13 | Guwahati | 0361-2337700 |
| 14 | Hisar | 01662-275787 |
| 15 | Hyderabad | 040-24581322 |
| 16 | Imphal | 0385-241644 |
| 17 | Jaipur | 0141 2550229 |
| 18 | Indore | 0731-2476188 |
| 19 | Jabalpur | 0761-2353138 |
| 20 | Jammu | 0191-2262012 |
| 21 | Karnal | 0184-2252800 |
| 22 | Kolkata | 033- 4212115 |
| 23 | Kota | 0744-2462642 |
| 24 | Lucknow | 0522-2480726 |
| 25 | Ludhiana | 0161-2308669 |
| 26 | Mannuthy | 0487-2370344 |
| 27 | Mohanpur | 033-25878338 |
| 29 | Nagpur | 07103-275536 |
| 30 | Palampur | 01894-230408 |
| 31 | Pantnagar | 05944-233407 |
| 32 | Patna | 0612- 2228882 |
| 33 | Port Blair | 03192-250436 |
| 34 | Pune | 020-25538489 |
| 35 | Pusa (Bihar) | 06274-240239 |
| 36 | Raipur | 0771-2442491 |
| 37 | Ranchi | 0651-2261156 |
| 38 | Shillong | 0364-2570257 |
| 39 | Solan | 01792-252219 |
| 40 | Srinagar (Kashmir) |
0194-2305044 |
| 41 | Tirupati | 0877-2249220 |
| 42 | Thiruvananthapuram | 0471-2598431 |
| 43 | Udaipur | 0294-2418976 |
| 44 | Varanasi | 0542-2635236 |
| 45 | Mathura | 0565-2471706 |
| 46 | Bareilly | 0581-2301584 |
| 47 | Jodhpur | 0291-2786584 |
| 48 | Kozhikode | 0496-2249371 |
| 49 | Parbhani | 02452-233007 |
| 50 | Jhalawar | 07432-24115507432-242155 |
AIEEA UG 2015 Important Dates

Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
The Indian Council of Agricultural Research (ICAR) AIEEA important Dates
ICAR AIEEA UG 2015 Important Dates
| Last date for receipt of application | 23rd February 2015 |
| Last date for receipt of applications from certain remote areas namely, Assam, Meghalaya, Arunachal Pradesh, Mizoram, Manipur, Nagaland, Tripura, Sikkim, Jammu & Kashmir, Lahaul & Spiti district and Pangi sub-division of Chamba district of Himachal Pradesh, Andaman & Nicobar Islands and Lakshadweep | 27th February 2015 |
| Admit Card or Roll No. query if any | After last week of March 2015 |
| Date and Time of Examination | Saturday, April 11 , 2015 (10.00 AM to 12.30 PM) |
| Declaration of Result | After 3rd week of May 2015 |
| Date of counseling schedule (Tentative) | 16th June 2015 – 26th June 2015 |
AIEEA UG 2015 Agriculture Syllabus

Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1: Agrometeorology, Genetics and Plant Breeding, Biochemistry and Microbiology
Agrometerology: Elements of Weather-rainfall, temperature, humidity, wind velocity, Sunshine weather forecasting, climate change in relation to crop production.
Genetics & Plants Breeding : (a) Cell and its structure, cell division-mitosis and meiosis and their significance (b) Organisation of the genetic materials in chromosomes, DNA and RNA (c) Mendel’s laws of inheritance. Reasons for the success of Mendel in his experiments, Absence of linkage in Mendel’s experiments. (d) Quantitative inheritance, continuous and discontinuous variation in plants. (e) Monogenic and polygenic inheritance. (f) Role of Genetics in Plant breeding, self and cross-pollinated crops, methods of breeding in field crops-introduction, selection, hybridization, mutation and polyploidy, tissue and cell culture, (g) Plant Biotechnology-definition and scope in crop production.
Biochemistry: pH and buffers, Classification and nomenclature of carbohydrates; proteins; lipids; vitamins and enzymes.
Microbiology: Microbial cell structure, Micro-organisms-Algae, Bacteria, Fungi, Actinomycetes, Protozoa and Viruses Role of micro-organisms in respiration, fermentation and organic matter decomposition.
Unit-2: Livestock Production
Scope and importance: (a) Importance of livestock in agriculture and industry. White revolution in India. (b) Important breeds Indian and exotic, distribution of cows, buffaloes and poultry in India.
Care and management: (a) Systems of cattle and poultry housing (b) Principles of feeding, feeding practices. (c) Balanced ration-definition and ingredients. (d) Management of calves, bullocks, pregnant and milch animals as well as chicks crockrels and layers, poultry. (e) Signs of sick animals, symptoms of common diseases in cattle and poultry, Rinderpest, black quarter, foot and mouth, mastitis and haemorrhagic septicaemia coccidiosis, Fowl pox and Ranikhet disease, their prevention and control.
Artificial Insemination: Reproductive organs, collection, dilution and preservation of semen and artificial insemination,
role of artificial insemination in cattle improvement, Livestock Products: Processing and marketing of milk and Milk products.
Unit-3: Crop Production
Introduction: (a) Targets and achievements in foodgrain production in India since independence and its future projections, sustainable crop production, commercialization of agriculture and its scope in India. (b) Classification of field crops based on their utility-cereals, pulses, oils seeds, fibre, sugar and forage crops.
Soil, Soil fertility, Fertilizers and Manures: (a) Soil, soil pH, Soil structure, soil organisms, soil organisms, soil tilth, soil fertility and soil health. (b) Essential plant nutrients, their functions and deficiency symptoms. (c) soil types of India and their characteristics. (d) Organic manure, common fertilizers including straight, complex, fertilizer mixtures and biofertilizers; integrated nutrient management system.
Irrigation and Drainage: (a) Sources of irrigation (rain, canals, rivers, wells, tubewells). (b) Scheduling of irrigation based on critical stages of growth, time interval, soil moisture content and weather parameters. (c) Water requirement of crops. (d) Methods of irrigation and drainage. (e) Watershed management.
Weed Control: Principles of weed control, methods of weed control (cultural, mechanical, chemical, biological and Integrated weed management)
Crops: Seed bed preparation, seed treatment, time and method of sowing/planting, seed rate; dose, method and time of fertilizer application, irrigation, interculture and weed control; common pests and diseases, caused by bacteria, fungi virus and nematode and their control, integrated pest management, harvesting, threshing, post harvest technology: storage, processing and marketing of major field crops-Rice, wheat, maize, sorghum, pearl millet, groundnut, mustard, pigeon-pea, gram, sugarcane, cotton and berseem.
Unit-4: Horticulture
(a) Importance of fruits and vegetables in human diet, Crop diversification & processing Industry. (b) orchard-location and layout, ornamental gardening and kitchen garden. (c) Planting system, training, pruning, intercropping, protection from frost and sunburn. (d) Trees, shrubs, climbers, annuals, perennials-definition and examples. Propagation by seed, cutting, budding, layering and grafting.(e) Cultivation practices, processing and marketing of: (i) Fruits – mango, papaya, banana, guava, citrus, grapes, (ii) Vegetables – Radish, Carrot, potato, onion, cauliflower, brinjal, tomato, spinach and cabbage, (iii) Flowers – Gladiolus, canna, chrysanthemus, roses and marigold. (f) Principles and methods of fruit and vegetable preservation. (g) Preparation of jellies, jams, ketchup, chips and their packing.
AIEEA UG 2015 Mathematics Syllabus

Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1: Sets and functions
1. Sets : Sets and their representations. Empty set. Finite & Infinite sets. Equal sets. Subsets, Subsets of the set of real numbers especially intervals (with notations). Power set. Universal set. Venn diagrams. Union and intersection of sets. Difference of sets. Complement of a set.
2. Relations & Functions : Ordered pairs, Cartesian product of sets. Number of elements in the Cartesian product of two finite sets. Cartesian product of the reals with itself (Upto R × R × R). Definition of relation, Types of relations: reflexive, symmetric, transitive and equivalence relations. One to one and onto functions, composite functions, inverse of a function. Binary operations. Pictorial representation of a function, domain. Co-domain and range of a relation. Function as a special kind of relation from one set to another. Real valued function of the real variable, domain and range of these functions, constant, identity, polynomial, rational, modulus, signum and greatest integer functions with their graphs. Sum, difference, product and quotients of functions.
3. Trigonometric Functions: Positive and negative angles. Measuring angles in radians & in degrees and conversion from one measure to another. Definition of trigonometric functions with the help of unit circle. Truth of the identity sin2x + cos2x = 1, for all x. Signs of trigonometric functions and sketch of their graphs. Expressing sin(x +y) and cos (x+y) in terms of sinx, siny, cosx & cosy. Deducing the identities like the following:
Identities related to sin2x, tan2x, sin3x, cos3x and tan3x. General solution of trigonometric equations of the type sin è ? = sin á, cos è ? = cos á? and tan é ? = tan á.
Inverse Trigonometric Functions: Definition, range, domain, principal value branches. Graphs of inverse trigonometric functions. Elementary properties of inverse trigonometric functions.
Properties of triangles, including centroid, incentre, circum-centre and orthocenter, Solution of triangle, Heights and Distances.
Unit-2: Algebra
1. Principle of Mathematical induction: Processes of the proof by induction, motivating the application of the method by looking at natural numbers as the least inductive subset of real numbers. The principle of mathematical induction and simple applications.
2. Complex Numbers and Quadratic Equations: Need for complex numbers, especially-1, to be motivated inability to solve every quadratic equation. Brief description of algebraic properties of complex numbers. Argand plane and polar representation of complex numbers. Statement of Fundamental Theorem of Algebra, solution of quadratic equations in the complex number system.
3. Linear Inequalities: Linear inequalities, Algebraic solutions of linear inequalities in one variable and their representation on the number line. Graphical solution of linear inequalities in two variables. Solution of system of linear inequalities in two variables- graphically.
4. Permutations & Combinations: Fundamental principle of counting. Factorial n.(n!). Permutations and combinations, derivation of formula and their connections, simple applications.
5. Binomial Theorem: History, statement and proof of the binomial theorem for positive integral indices. pascal’s triangle, General and middle term in binomial expansion, simple applications.
6. Sequence and Series: Sequence and Series, Arithematic progression (A.P.). arithmetic mean (A.M.) Geometric progression(G.P), general term of G.P., sum of n terms of a G.P., geometric mean(G.M.). relation between A.M. and G.M. Sum to ne terms of the special series Ón, Ón2, and Ón3.
7. Matrices: Concept, notation, order, equality, types of matrices, zero matrix, transpose of a matrix, symmetric and skew symmetric matrices. Addition, multiplication and scalar multiplication of matrices, simple properties of addition, multiplication and scalar multiplication. None-commutativity of multiplication of matrices and existence of non-zero matrices whose product is the zero matrix(restrict to square matrices of order 2). Concept of elementary row and column operations. Invertible matrices and proof of the unquencess of inverse, if it exists.
8. Determinants: Determinant of a square matrix( up to 3 × 3 matrices), properties of determinants, minors, cofactors and application of determinants in finding the area of a triangle. Adjoint and inverse of a square matrix. Consistency, inconsistency and number of solutions of system of linear equation by examples, solving system of linear equations in two or three variables (having unique solution) using inverse of a matrix.
Unit-3: Coordinate Geometry
1. Straight Lines: Slope of a line and angle between two lines. Various forms of equations of a line: parallel to axes, point-slope form, slope-intercept form, two-point form, intercepts form and normal form. General equation of a line. Distance of a point from a line.
2. Conic Sections: Sections of a cone: circle ellipse, parabola, hyperbola, a point, a straight line and pair of parabola, ellipse and hyperbola. Standard equation of a circle.
3. Introduction to Three-dimensional Geometry: Coordinate axes and coordinate planes in three dimensions. Coordinates of a point. Distance between two points and section formula.
Unit-4: Calculus
1. Limits and Derivatives: Derivative introduced as rate of change both as that of distance function and geometrically, intuitive idea of limit. Definition of derivative, relate it to slope of tangent of the curve, derivative of sum, difference, product and quotient of functions. Derivatives of polynomial and trigonometric functions.
2. Continuity and Differentiability: Continuity and differentiability, derivative of composite functions, chain rule, derivatives of inverse trigonometric functions, derivative of implicit function. Concept of exponential and logarithmic functions and their derivative. Logarithmic differentiation. derivative of functions expressed in parametric forms. Second order derivatives. Rolle’s and Lagrang’s Mean Value Theorems (without proof) and their geometric interpretations.
3. Application of Derivatives: Application of derivatives: rate of change, increasing/decreasing functions, tangents & normals, approximation, maxima and minima(first derivative test motivated geometrically and second derivative test given as a provable tool). Sample problems.
4. Integrals: Integration as inverse process of differentiation. Integration of a variety of functions by substitution, by partial fractions and by parts; only simple integrals of the type
to be a evaluated. Definite integrals as a limit of a sum, Fundamental Theorem of Calculus (without proof). Basic properties of definite integrals and evaluation of definite integrals.
5. Applications of the Integrals : Application in finding the areas under simple curves, especially lines, areas of circles/parabolas/ellipses(in standard form only), area between the two above said curves.
6. Differential Equations: Definition, order and degree, general and particular solutions of a differential equation, Formation of differential equation whose general solution is given. Solution of differential equations by method of separation of variables, homogenous differential equations of first order and first degree. Solutions of linear differential equation of the type: ![]() where p and q are function of x.
where p and q are function of x.
Unit-5: Vectors and Three-Dimensional Geometry
1. Vectors: Vectors and scalars, magnitude and direction of a vector. Direction cosines/ratios of vectors. Types of vectors (equal unit, zero, parallel and collinear vectors), position vector of a pint, negative of a vector, components of a vector, addition of vectors, multiplication of a vector by a scalar position vector of a point dividing a line segment in a given ratio. Scalar (dot) product of vectors, projection of a vector on a line. Vector(cross) product of vectors.
2. Three-dimensional Geometry: Direction cosines/ratios of a line joining two points. Cartesian and vector equation of a line, coplanar and skew lines, shortest distance between two lines. Cartesian and vector equation of a plane. Angle between(i) two lines, (ii) two planes. (iii) a line and a plane. Distance of a point from a plane.
Unit-6: Linear Programming
Linear Programming: Introduction, definition of related terminology such as constraints, objective function, optimization, different types of linear programming (L.P.) problems, mathematical formulation of L.P. problems, graphical method of solution for problems in two variables, feasible and infeasible regions, feasible and infeasible solutions, optimal feasible solutions (up to three non-trivial constraints).
Unit-7: Mathematical Reasoning
Mathematical Reasoning: Mathematically acceptable statements. Connecting words/phrases – consolidating the understanding of “if and only if (necessary and sufficient) condition”, “implies”, “and/or”, “implied by”, “and” “or”, “there exists” and their use through variety of examples related to real life and Mathematics Validating the statements involving the connecting words, difference between contradiction, converse and contra positive.
Unit-8: Statistics & Probability
1. Statistics: Measures of central tendency, mean, median and mode from ungrouped/grouped data. Measures of dispersion, mean deviation, variance and standard deviation from ungrouped/ data. Correlation, regression lines.
2. Probability: Random experiments: outcomes, sample spaces (set representation) Events: occurrence of events, ‘not’, ‘and’ and ‘or’ events, exhaustive events, mutually exclusive events Axiomatic (set theoretic) probability, Probability of an event, probability of ‘not’, ‘and’ & ‘or’ events. Multiplication theorem on probability. Conditional probability, independent events, total probability. Bayes’ theorem, Random variable and its probability distribution, mean and variance of stochastic variable. Repeated independent (Bernoulli) trials and Binomial distribution.
Unit-9: Statics
Introduction: basic concepts and basic laws of mechanics, force resultant of forces acting at a point, parallelogram law of forces, resolved parts of a force, Equilibrium of a particle under three concurrent forces. Triangle law of forces and its converse, Lami’s theorem and its converse, Two parallel forces, like and unlike parallel forces, couple and its moment.
Unit-10: Dynamics
Speed and velocity, average speed, instantaneous speed, acceleration and retardation, resultant of two velocities. Motion of a particle along a line, moving with constant acceleration. Motion under gravity. Laws of motion, Projectile motion.
AIEEA UG 2015 Biology Syllabus

Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit: 1 The living World
Nature and scope of Biology, Methods of Biology. Our place in universe. Laws that govern the universe and life. Level of organization. Cause and effect relationship.
Origin of life and its maintenance. Origin and diversity of life. Physical and chemical principles that maintain life processes. The living crust and interdependence. The positive and negative aspects of progress in biological sciences. The future of the living world. identification of human responsibility in shaping our future.
Unit:2 Unit of Life
Cells as a unit of life. Small biomolecules; water, minerals, mono and oligosaccharides, lipids, amino acids, nucleotides and their chemistry, cellular location and function. Macromolecules in cells – their chemistry, cellular location and functional significance, Polysaccharides, proteins and nucleic acids. Enzymes; chemical nature, classification, mechanism in action-enzyme complex, allosteric modulation (brief), inrreversible activation. Biomembranes; Fluid mosaic model of membrane, role in transport, recognition of external information (brief). Structural organization of the cell; light and electron microscopic view of cell, its organelles and their functions; nucleus mitochondria, chloroplasts, endoplasmic reticulum. Golgi complex, lysosomes, microtubules, cell wall, cilia and flagella, vacuoles, cell inclusions. A general account of cellular respiration. Fermentation, biological oxidation (A cycle outline), mitochondrial electron transports chain, high energy bonds and oxidative phosphorylation, cell reproduction; process of mitosis and meiosis.
Unit-3: Diversity of Life
Introduction. The enormous variety of living things, the need for classification to cope with this variety; taxonomy and phylogeny; shortcomings of a two kingdom classification as plants and animals; the five kingdom classification, Monera, Protista, Plantae, Fungi and Animalia; the basic features of five kingdom classification. Modes of obtaining nutrition-autotrophs and heterotrophs. Life style producers, consumers and decomposers. Unicellularity and multicellularity, phyylogenetic relationships. Concepts of species, taxon and categories – hierarchical levels of classification; binomial nomenclature; principles of classification and nomenclature; identification and nature of viruses and bacteriophages; kingdom Monera-archeabacteria – life in extermen environments; Bacteria, Atinomycetes, Cyanobacteria. Examples & illustration of autotrophic and heterotrophic life; mineralizes-nitrogen fixers; Monera in cycling matter; symbiotic forms; disease producers. Kingdom Protissta-Eukaryotic unicellular organisms, development of flagella and cilia; beginning of mitosis; syngamy and sex. Various life styles shown in the major phyla. Evolutionary precursors of complex life forms. Diatoms, dinoflagellates, slime moulds, protozons; symbiotic forms. Plant kingdom-complex autotrophs, red brown and green algae; conquest of land, bryophytes, ferns, gymnosperms and angiosperms. Vascularization; development of flower, fruit and seed. Kingdom fungi-lower fungi(Zygomycetes), higher fungi (Ascomycetes and Basidiomycetes); the importance o fungi. Decomposers; parasitic forms; lichens and mycorrhizae. Animal kingdom-animal body pattern and symmetry. The development of body cavity in invertebrate vertebrate physia. Salient features with reference to habitat and example of phylum porifera, coelenterate, helminthes, annelids, mollusca, arthropoda, echinoderms; chordate (classes-fishes, amphibians, reptiles, birds and mammals) highlighting major characters.
Unit:4 Organisms and Environment
Species : Origin and concept of species population, interaction between environment and population community. Biotic community, interaction between different species, biotic stability. Changes in the community. Succession, Ecosystem; interaction between biotic and abiotic components; major ecosystems, manmade ecosystem – Agro ecosystem. Biosphere; flow of energy, trapping of solar energy, energy pathway, food web, biogeochemial cycles, calcium and sulphur, ecological imbalance and its consequences. Conservation of natural resources’ nerewable and non-renewable (in brief). Water and land management, wasteland development. Wild life and forest conservation; causes for the extinction of some wild life, steps taken to conserve the remaining species, concept of endangered concept of afforestation. Environmental pollution; air and water pollution, sources, major pollutants of big cities of our country, their effects and methods of control, pollution due to fallout and waste disposal, effect and control, noise pollution; sources and effects.
Unit:5 Multicellulairty : Structure and Function – Plant Life
Form and function. Tissue system in flowering plants; meristematic and permanent. Mineral nutrition-essential elements, major functions of different elements, passive and active uptake of minerals. Modes of nutrition, transport of solutes and water in plants. Photosynthesis; photochemical and biosynthetic phases, diversity in photosynthetic pathways, photosynthetic electron transport and photophosphorylation, photorespiration. Transpiration and exchange of gases. Stomatal mechanism. Osmoregulation in plants: water relations in plant cells, water potential. Reproduction and development in Angiosperms; asexual and sexual reproduction. Structure and functions of flower : development of male and female gametophytes in angiosperms, pollination, fertilization and development of endosperm, embryo seed and fruit. Differentiation and organ formation. Plant hormones and growth regulation; action of plant hormones in relation to seed dormancy and germination, apical dominance. senescence and abscission. Application of synthetic growth regulators. A brief account of growth and movement in plants.
Unit:6 Multicellularity : Structure and Function – Animal Life
Animal tissues, epithelial, connective, muscular, nerve. Animal nutrition, organs of digestion and digestive process, nutritional requirements for carbohydrates, proteins, fats, mineral and vitamins; nutritional imbalances and deficiency diseases. Gas exchange and transport: Pulmonary gas exchange and organs involved, transport of gases in blood, gas exchange in aqueous medic circulation: closed and open vascular systems, structure and pumping action of heart, arterial blood pressure, lymph. Excretion and osomoregulation. Ammonotelism, Ureotelism, urecotelism, excretion of water and urea with special reference to man. Role of kidney in regulation of plasma, osmolarity on the basis of nephron structure, skin and lungs in excretion. Hormonal coordination; hormones of mammals, role of hormones as messengers and regulators. Nervous coordination, central autonomic and peripheral nervous systems, receptors, effectors, reflex action, basic physiology of special senses, integrative control by neuroendocrinal systems. Locomotion: joints, muscle movements, types of skeletal muscles according to types of movement, basic aspects of human skeleton. Reproduction; human reproduction, female reproductive cycle. Embryonic development in mammals (upto three germs layers), growth, repair and ageing.
Unit:7 Continuity of Life
Heredity and variation: Introduction, Mendel’s experiments with peas and concepts of factors. Mendel’s laws of inheritance. Genes: Packaging of heredity material in prokaryotes-bacterial chromosome and plasmid; and eukaryote chromosomes. Extranuclear genes, viral genes. Linkage (genetic) maps. Sex determination and sex linkage. Genetic material and its replication, gene manipulation, Gene expression; genetic code, transcription, translation, gene regulation. Molecular basis of differentiation.
Unit:8 Origin and Evolution of Life
Origin of life: living and non-living, chemical evolution. organic evolution; Oparin ideas, Miller-Urey experiments. Interrelationship among living organisms and evidences of evolution: fossil records including geological scale, Morphological evidence – hematology, vestigial organs, embryological similarities and biogeographical evidence.
Darwin’s two major contributoions. Common origin of living organisms and recombination as sourece of variability selection and variation, adaptation (Lederberg’s replica plating experiment for indirect selection of bacterial mutants), reproductive isolation, speciation. Role of selection, change and drift in determining composition of population. Selected examples: industrial melanism; drug resistance, mimicry, malaria in relation to G-6-PD deficiency and sickle cell disease. Human evolution: Placontological evidence, man’s place among mammals. Brief idea of dryopithecus, Australopithecus, Homo erectus. H. neanderthlensis, Cro-Magnon man and Homo sapiens. Human chromosomes, similarity in different racial groups. Comparison with chromosomes of non-human primates to indicate common origin; Cultural vs. biological evolution.
Mutation: origin and types of mutation, their role in speciation.
Unit:9 Application of Biology
Introduction, role of biology, in the amelioration of human problems. Domestication of plant- a historical account, improvement of crop plants; Principles of plant breeding and plant introduction. Use of fertilizers, their economic and ecological aspects.
Use of pesticides: advantages and hazards. Biological methods of pest control. Crops today. Current concerns, gene pools and genetic conservation. Underultilized crops with potential uses of oilseeds, medicines, beverages, spices, fodder. New crops-Leucaena (Subabul), Jojoba, Guayule, winged bean, etc. Biofertilizers – green manure, crop residues and nitrogen fixation (symbiotic, non symbiotic). Applications of tissue culture and genetic engineering in crops. Domestication and introduction of animals. Livestock, poultry, fisheries (Fresh water, marine, aquaculture). Improvement of animals: principles of animal breeding. Major animal diseases and their control. Insects and their products (silk, honey, wax and lac). Bioenergy-biomass, wood(combustion; gasification, ethanol). Cow dung cakes, gobar gas, plants as sources of hydrocarbons, for producing petroleum, ethanol from starch and lignocelluloses. Biotechnology, application in health and agriculture, genetically modified (GM) organisms, bio-safety issues. A brief historical account-manufacture of cheese. yoghurt, alcohol, yeast, vitamins, organic acids, antibiotics, steroids, dextrins. Scalling up laboratory findings to industrial production, sewage treatment. Production in insulin, human growth hormones, interferon. Communicable diseases including STD and diseases spread through blood transfusion (hepatitis, AIDS, etc) Immune response, vaccine and antisera. Allergies and Inflammation. Inherited diseases and dysfunction, sex-linked diseases, genetic incompatibilities, and genetic counseling. Cancer-major types. causes, diagnosis and treatment. Tissue and organ transplantation. Community health services and measures; blood banks; mental health, smoking, alcoholism and drug addiction-physiological symptoms and control measures. Industrial wastes, toxicology, pollution-related diseases. Biomedical engineering – spare parts for man, instruments for diagnosis of diseases and care. Human population related diseases. Human population, growth, problems and control, inequality between sexes, control measures; test-tube babies aminocentesis. Future of Biology.
AIEEA UG 2015 Chemistry Syllabus

Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1 : Some Basic Concepts of Chemistry
General Introduction: Importance and scope of chemistry. Historical approach to particulate nature of matter, laws of chemical combination. Dalton’s atomic theory: concept of elements, atoms and molecules. Atomic and molecular masses mole concept and molar mass: percentage composition, empirical and molecular formula chemical reactions, stoichiometry and calculations based on stoichiometry.
Unit-2: Solid State
Classification of solid based on different binding forces : molecular, ionic, covalent and metallic solids, amorphous and crystalline solids (elementary idea), unit cell in two dimensional and three dimensional lattices, calculation of density of unit cell, packing in solids, voids, number of atoms per unit cell in a cubic unit cell, point defects, electrical and magnetic properties.
Unit-3: Solutions
Types of solutions, expression of concentration of solutions of solids in liquids, solubility of gases in liquids, solid solutions, colligative properties-relative lowering of vapour pressure, elevation of Boiling Point, depression of freezing point, osmotic pressure, determination of molecular masses using colligative properties, abnormal molecular mass.
Unit-4: Structure of Atom
Discovery of electron, proton and neutron; atomic number, isotopes and isobars. Thomson’s model and its limitations, Rutherford’s model and its limitations. Bohr’s model and its limitations, concept of shells and subshells, dual nature of matter and light, de Broglie’s relationship. Heisenberg uncertainty principle, concept of orbitals, quantum numbers, shapes of s, p, and d orbitals, rules for filling electrons in orbitals-Aufabau principle, Pauli exclusion principle and Hund’s rule, electronic configuration of atoms, stability of half filled and completely filled orbitals.
Unit-5: Classification of Elements and Periodicity in Properties
Significance of classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements –atomic radii, ionic radii. Ionization enthalpy, electron gain enthalpy, electro negativity, valence.
Unit-6: Chemical Bonding and Molecular Structure
Valence electrons, ionic bond, covalent bond: bond parameters. Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR(Valence shell electron pair repulsion) theory, concept of hybridization, involving s, p and d orbitals and shapes of some simple molecules, molecular orbitas; theory of homonuclear diatomic molecules (qualitative idea only), hydrogen bond.
Unit-7: States of Matter: Gases and Liquids
Three states of matte. Intermolecular interactions, type of bonding, melting and boiling points. Role of gas laws in elucidating the concept of the molecule, Boyle’s law. Charles law, Gay Lussac’s law, Avogadro’s law. Ideal behavior, empirical derivation of gas equation, Avogadro’s number. Ideal gas equation. Derivation from ideal behavior, liquefaction of gases, critical temperature. Liquid State- Vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations)
Unit-8: Thermodynamics
Concepts of System, types of systems, surroundings, Work, heat, energy, extensive and intensive properties, state functions. First law of thermodynamics – internal energy and enthalpy, heat capacity and specific heat, measurement of DU and DH, Hess’s law of constant heat summation, enthalpy of: bond dissociation, combustion, formation, atomization, sublimation. Phase transformation, ionization, and solution. Introduction of entropy as a state function, free energy change for spontaneous and non-spontaneous processes, criteria for equilibrium.
Unit-9: Equilibrium
Equilibrium in physical and chemical processes, dynamic nature of equilibrium, law of mass of action, equilibrium constant, factors affecting equilibrium – Le Chatelier’s principle; ionic equilibrium – ionization of acids and bases, strong and weak electrolytes, degree of ionization, concept of pH. Hydrolysis of salts. Buffer solutions, solubility product, common ion effect.
Unit-10: Redox Reactions
Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, applications of redox reactions.
Unit-11: Hydrogen
Position of hydrogen in periodic table, occurrence, isotopes, preparation, properties and uses of hydrogen; hydrides – ionic, covalent and interstitial; physical and chemical properties of water, heavy water; hydrogen peroxide-preparation, properties and structure; hydrogen as a fuel.
Unit-12: s-Block Elements (Alkali and Alkaline earth metals)
Group 1 and Group 2 elements
General introduction, electronic configuration, occurrence, anomalous properties of the first element each group, diagonal relationship, trends in the variation of properties (such as ionization enthalpy, atomic and ionic radii), trends in chemical reactivity with oxygen, water, hydrogen and halogens; uses.
Unit-13: Preparation and properties of some important compounds
Sodium carbonate, sodium chloride, sodium hydroxide and sodium hydrogen carbonate, biological importance of sodium and potassium. CaO, CaCO3 and industrial use of lime and limestone, biological importance of Mg and Ca
Unit-14: Some p-Block Elements
General Introduction to p-Block Elements: Group 13 elements
General introduction, electronic configuration, occurrence. Variation of properties, oxidations states, trends in chemical reactivity, anomalous properties of first element of the group; Boron-physical and chemical properties, some important compound: borax, boric acids, boron hydrides. Aluminum: uses, reactions with acids and alkalies.
Unit-15: Group 14 elements
General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous behavior of first element, Carbon – catenation, allotropic forms, physical and chemical properties; uses of some important compounds: oxides. Important compounds of silicon and a few uses: silicon tetrachloride, silicones, silicates and zeolites.
Unit-16: Organic Chemistry
Some Basic Principles and Techniques
General Introduction, methods of qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds. Electronic displacements in a covalent bond: inductive effect, electromeric effect, resonance and hyper conjugation. Homolytic and heterolytic fission of a covalent bond: free radicals, carbocations, carbanions; electrophiles and nucleophiles, types of organic reactions.
Unit-17: Hydrocarbons
Classification of hydrocarbons
Alkanes – Nomenclature, isomerism, conformations (ethane only), physical properties, chemical reactions including free radical mechanism of halogenation, combustion and pyrolysis.
Alkenes – Nomenclature, structure of double bond (ethane) geometrical isomerism, physical properties, methods of preparation; chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikov’s addition and peroxide effect), ozonolysis, oxidation, mechanism of electrophilic addition.
Alkynes – Nomenclature, structure of triple bind (ethyne), physical properties. Methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of – hydrogen, halogens, hydrogen halides and water.
Aromatic hydrocarbons: Introduction, IUPAC nomenclature; benzene: resonance, aromaticity, chemical properties mechanism of electrophilic substitution. – nitration, sulphonation, halogenations. Friedel-Craft’s alkuylation and acylation: directive influence of functional group in mono-substituted benzene; carcinogenicity and toxicity.
Unit-18: Electrochemistry
Conductance in electrolytic solutions, specific and molar conductivity variations of conductivity with concentration, Kohlrausch’s Law, electrolysis and laws of electrolysis (elementary idea), dry cell – electrolytic cells and Galvanic cells: lead accumulator, EMF of a cell, standard electrode potential, Nernst equation and its application to chemical cells, fuel cells; corrosion.
Unit-19: Chemical Kinetics
Rate of a reaction (average and instantaneous), factors affecting rate of reaction; concentration, temperature, catalyst; order and molecularity of a reaction; rate law and specific rate constant, integrated rate equations and half life (only for zero and first order reactions); concept of collision theory (elementary idea, no mathematical treatment)
Unit-20: Surface Chemistry
Adsoprtion – physisorption and chemisorptions; factors affecting adsorption of gases on solids; catalysis : homogenous and heterogeneous, activity and selectivity: enzyme catalysis; colloidal state: distinction between true solutions, colloids and suspensions; lyophilic lyophobic, multimolecular and macromolecular colloids; properties of colloids; Tyndall effect, Brownian movement, electrophoresis, coagulation; emulsion – types of emulsions.
Unit-21: General Principles and Processes of Isolation of Elements
Principles and methods of extraction – concentration, oxidation, reduction electrolytic method and refining; occurrence and principles of extraction of aluminium, copper, zinc and iron.
Unit-22: p-Block Elements
Group 15 elements
General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; nitrogen – preparation, properties and uses; compounds of nitrogen: preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous-allotropic froms; compounds of phosphorous: preparation and properties of phosphine, halides(PCl3, PCl5) and oxoacids
Unit-23: Group 16 elements
General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; simple oxides; Ozone. Sulphur-allotropic forms; compounds of sulphur: preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Unit-24: Group 17 elements
General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds, oxoacids of halogens (structures only).
Unit-25: Group 18 elements
General introduction, electronic configuration. Occurrence, trends in physical and chemical properties, uses.
Unit-26: d and f Block Elements
General introduction, electronic configuration, occurrence and characteristics of transition metals. General trends in properties of the first row transition metals – metallic character, ionization enthalpy, oxidation states, ionic radii, colour catalytic property, magnetic properties, interstitial compounds, alloy formation preparation and properties of K2Cr2O7 and KMnO4.
Lanthnoids – electronic configuration, oxidation states, chemical reactivity and lanthanoid contraction.
Actinoids – Electronic configuration, oxidation states.
Unit-27: Coordination Compounds
Coordination compounds – Introduction, ligands, coordination number, colour, magnetic properties and shapes, IUPAC nomenclature of mononuclear coordination compounds, bonding; isomerism, importance of coordination compounds (in qualitative analysis, extraction of metals and biological systems).
Unit-28: Haloalkanes and Haloarenes
Halokanes : Nomenclature, nature of C-X bond, physical and chemical properties, mechanism of substitution reactions.
Halorenes : Nature of C-X bond, substitution reactions (directive influence of halogen for monosubstituted compounds only) Uses and environmental effects of – dichloromethane, trichloromethane, tetrachloromethane, iodoform, freons, DDT.
Unit-29: Alcohols, Phenols and Ethers
Alcohols
Nomenclature, methods of preparation, physical and chemical properties (of primary alcohols only); identification of primary, secondary and tertiary alcohols; mechanism of dehydration, uses of methanol and ethanol. Phenols : Nomenclature, methods of preparation, physical and chemical properties, acidic nature of phenol, electrophillic substitution reactions, uses of phenols, Ethers: Nomenclature, methods of preparation, physical and chemical properties, uses,
Unit-30: Aldehydes, Ketones and Carboxylic Acids
Aldehydes and Ketones: Nomenclature, nature of carbonyl group, methods of preparation, physical and chemical properties mechanism of nucleophillic addition, reactivity of alpha hydrogen in aldehydes; uses.
Carboxylic Acids: Nomenclature, acidic nature, methods of preparation, physical and chemical properties; uses.
Unit-31: Organic compounds containing Nitrogen
Amines: Nomenclature, classification, structure, methods of preparation, physical and chemical properties, uses, identification of primary, secondary and tertiary amines.
Cyanides and Isocyanides – will be mentioned at relevant places in context.
Diazonium salts: Preparation, chemical reactions and importance in synthetic organic chemistry.
Unit-32: Biomolecules
Carbohydrates – Classification (aldoses and ketoses), monosaccharide (glucose and fructose), oligosaccharides (sucrose, lactose, maltose), polysaccharides (starch, cellulose, glycogen); importance
Proteins – Elementary idea of a-amino acids, peptide bond, polypeptides, proteins, structure of amines-primary, secondary, tertiary structure and quaternary structures (qualitative idea only), denaturation of proteins; enzymes.
Vitamins – Classification and functions.
Nucleic Acids: DNA and RNA.
Unit-33: Polymers
Classification- natural and synthetic, methods of polymerization (addition and condensation), copolymerization. Some important polymers: natural and synthetic like polythene, nylon, polyesters, Bakelite, rubber.
Unit-34: Environmental Chemistry
Environmental pollution – air,, water and soil pollution, chemical reactions in atmosphere, smog, major atmospheric pollutants; acids rain, ozone and its reactions, effects of depletion of ozone layer, greenhouse effect and global warming – pollution due to industrial wastes; green chemistry as an alternative tool for reducing pollution, strategy for control of environmental pollution.
Unit-35: Chemistry in Everyday life
1. Chemical in Medicines – analgesics, tranquilizers, antiseptics, disinfectants, antimicrobials, antifertility drugs, antibiotics, antacids, antihistamines.
2. Chemical in food – preservatives, artificial sweetening agents.
3. Cleansing agents – soaps and detergents, cleansing action.
AIEEA UG 2015 Physics Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1 : Physical World and Measurement
Physics scope and excitement; nature of physical laws; Physics, technology and society, Need for measurement: Units of measurement; systems of units; SI nits, fundamental and derived units. Length, mass and time measurements; accuracy and precision of measuring instruments; errors in measurement; significant figures. Dimensions of physical quantities, dimensional analysis and its applications.
Unit-2: Kinematics
Frame of reference, Motion in a straight line: Position-time graph, speed and velocity. Uniform and non-uniform motion, average speed and instantaneous velocity. Uniformly accelerated motion; velocity-time graph, position-time graphs, relations for uniformly accelerated motion (graphical treatment). Elementary concepts of differentiation and integration for describing motion. Scalar and vector quantities: Position and displacement vectors, general vectors and notation, equality of vectors, multiplication of vectors by a real number; addition and subtraction of vectors. Relative velocity. Unit vector; Resolution of a vector in a plane – rectangular components. Motion in a plane. Cases of uniform velocity and uniform acceleration-projectile motion. Uniform circular motion. Motion of objects in three dimensional space. Motion of objects in three dimensional space.
Unit-3: Laws of Motion
Intuitive concept of force, Inertia, Newton’s first law of motion; momentum and Newton’s second law of motion; impulse; Newton’s third law of motion. Law of conservation of linear momentum and its applications. Equilibrium of concurrent forces, Static and kinetic friction, laws of friction, rolling friction. Dynamics of uniform circular motion:
Centripetal force, examples of circular motion(vehicle on level circular road, vehicle on banked road).
Unit-4: Work, Energy and Power
Scalar product of vectors. Work done by a constant force and a variable force; kinetic energy, work-energy, work-energy theorem, power. Notion of potential energy, potential energy of a spring, conservative forces: conservation of mechanical energy (Kinetic and potential energies); non-conservative forces: elastic and inelastic collisions in one and two dimensions)
Unit-5: Motion of System of Particles and Rigid Body
Centre of mass of a two-particle system, momentum conversation and centre of mass motion. Centre of mass of a rigid body; centre of mass of uniform rod. Vector product of vectors; moment of a force, torque, angular momentum, conservation of angular momentum with some examples. Equilibrium of rigid bodies, rigid body rotation and equations of rotational motion, comparison of linear and rotational motions; moment of inertia, radius of gyration. Values of moments of inertia for simple geometrical objects. Statement of parallel and perpendicular axes theorems and their applications.
Unit-6: Gravitation
Keplar’s laws of planetary motion. The universal law of gravitation. Acceleration due to gravity and its variation with altitude and depth. Gravitational potential energy; gravitational potential. Escape velocity. Orbital velocity a satellite. Geo-stationary satellites.
Unit-7: Properties of Bulk Matter
Elastic behavior, Stress-strain relationship, Hooke’s law, Young’s modulus, bulk modulus, shear, modulus of rigidity. Pressure due to a fluid column; Pascal’s law and its applications (hydraulic lift and hydraulic brakes). Effect of gravity on fluid pressure. Viscosity, Stokes’ law, terminal velocity, Reynold’s number, streamline and turbulent flow.
Bernoulli’s theorem and its applications. Surface energy and surface tension, angle of contact, application of surface tension ideas to drops, bubbles and capillary rise.
Heat, temperature, thermal expansion; specific heat-calorimetry; change of state-latent heat. Heat transfer-conduction, convection and radiation, thermal conductivity, Newton’s law of cooling.
Unit-8: Thermodynamics
Thermal equilibrium and definition of temperature (zeroth law of thermodynamics). Heat, work and internal energy, First law of thermodynamic. Second law of thermodynamics: reversible and irreversible processes. Heat engines and refrigerators.
Unit-9: Behaviour of Perfect Gas and Kinetic Theory
Equation of state of a perfect gas, work done on compressing a gas. Kinetic theory of gases – assumptions, concept of pressure. Kinetic energy and temperature; rms speed of gas molecules; degrees of freedom, law of equipartition of energy(statement only) and application to specific heats of gases; concept of mean free path, Avogadro’s number.
Unit-10: Oscillations and Waves
Periodic motion – period, frequency, displacement as a function of time. Periodic functions. Simple Harmonic Motion (S.H.M) and its equation; phase oscillations of a spring-restoring force and force constant; energy in S.H.M – kinetic and potential energies; simple pendulum – derivation of expression for its time period; free, forced and damped oscillations, resonance. Wave motion. Longitudinal and transverse waves. Speed of wave motion. Displacement relation for a progressive wave. Principle of superposition of waves, reflection of waves, standing waves in strings and organ pipes, fundamental mode and harmonics. Beats, Doppler effect.
Unit-11: Electrostatics
Electric Charges; Conservation of charge. Coulomb’s law – force between two point charges, forces between multiple charges; superposition
principle and continuous charge distribution. Electric field, electric field due to a point charge, electric field lines; electric dipole, electric field due to a dipole; torque on a dipole in uniform electric field. Electric fluid, statement of Gauss’s theorem and its applications to find field due to infinitely long straight wire, uniformly charged infinite plane sheet and uniformly charged thin spherical shell (field inside and outside). Electric potential, potential difference, electric potential due to a point charge, a dipole and system of charges; equipotential surfaces, electrical potential energy of a system of two point charges and of electric dipole in an electrostatic field. Conductors and insulators, free charges and bound charges inside a conductor, Dielectrics and electric polarization, capacitors and capacitances, combination of capacitors in series and in parallel, capacitance of a parallel plate capacitor with and without dielectric medium between the plates, energy stored in a capacitor. Van de Graff generator.
Unit-12: Current Electricity
Electric current, flow of electric charges in a metallic conductor, drift velocity, mobility and their relation with electric current; Ohm’s law, electrical resistance, V – I characteristics (linear and non-linear), electrical energy and power, electrical resistivity and conductivity. Carbon resistors, colour code for carbon resistors; series and parallel combinations of resistors; temperature dependence of resistance. Internal resistance of a cell, potential difference and emf of a cell, combination of cells in series and in parallel. Kirchoff’s laws and simple applications. Wheatstone bridge, metre bridge. Potentiometer – principle and its applications to measure potential difference and for comparing emf of two cells; measurement of internal resistance of a cell.
Unit-13: Magnetic Effects of Current and Magnetism
Concept of magnetic field, Oersted’s experiment. Biot-Savart law and its application to current carrying circular loop. Ampere’s alw and its applications to infinitely long straight wire, straight and toroidal solenoids. Force on a moving charge in uniform magnetic and electric fields.
Cyclotron. Force on a current-carrying conductor in a uniform magnetic field. Force between two parallel current-carrying conductors-definition of ampere. Torque experienced by a current loop in uniform magnetic field; moving coil galvanometer-its current sensitivity and conversion to ammeter and voltmeter.
Current loop as a magnetic dipole and its magnetic dipole moment. Magnetic dipole moment of a revolving electron. Magnetic field intensity due to a magnetic dipole (bar magnet) along its axis and perpendicular to its axis. Torque on a magnetic dipole (bar magnet) in a uniform magnetic field; bar magnet as an equivalent solenoid, magnetic field lines; Earth’s magnetic field and magnetic elements. Para-, dia- and ferro- magnetic substances, with examples Electromagnets and factors affecting their strengths. Permanent magnets.
Unit-14- Electromagnetic Induction and Alternating Currents
Electromagnetic induction; Faraday’s law, induced emf and current; Lenz’ Law, Eddy currents. Self and mutual inductance. Need for displacement current. Alternating currents, peak and rms value of alternating current/voltage; reactance and impedance; LC oscillations (qualitative treatment only). LCR series circuit, resonance; power in AC circuits, wattles current. AC generator and transformer.
Unit-15: Electromagnetic waves
Displacement current, Electromagnetic waves and their characteristics (qualitative ideas only). Transverse nature of electromagnetic waves. Electromagnetic spectrum (radio waves, microwaves, infrared, visible, ultraviolet, X-rays, gamma rays) including elementary facts about their uses.
Unit-16: Optics
Reflection of light, spherical mirrors, mirror formula. Refraction of light, total internal reflection and its applications, optical firbes, refraction at spherical surfaces, lenses, thin lens formula, lensmaker’s formula. Magnification, power of a lens, combination of thin lenses in contact.
Refraction and dispersion of light through a prism. Scattering of light – blue colour of the sky and reddish appearance of the sun at sunrise and sunset. Optical instruments: Human eye, image formation and accommodation. correction of eye defects (myopia, hypermetropia, presbyopia and astigmatism) using lenses. Microscopes and astronomical telescope (reflecting and refracting) and their magnifying powers. Wave optics: wave front and Huygens’ principle, reflection and refraction of plane wave at a plane surface using wave fronts. Proof of laws of reflection and refraction using Huygens’ principle. Interference, Young’s double slit experiment and expression for fringe width, coherent sources and sustained interference of light. Diffraction due to a single slit, width of central maximum. Resolving power of microscopes and astronomical telescopes. Polarization, plane polarized light; Brewster’s law, uses of p lane polarized light and Polaroids.
Unit-17: Dual Nature of Matter and Radiation
Dual nature of radiation. Photoelectric effect, Hertz and Lenard’s observations; Einstein’s photoelectric equation-particle nature of light. Matter waves-wave nature of particles, de Broglie relation. Davisson-Germer experiment.
Unit-18: Atoms & Nuclei
Alpha-particle scattering experiment; Rutherford’s model of atom; Bohr model, energy levels, hydrogen spectrum. Composition and size of nucleus, atomic masses, isotopes; isotones. Radioactivity, alpha, beta and gamma particles/rays and their properties; radioactive decay law. Mass-energy per nucleon and its variation with mass number; nuclear fission, nuclear reactor, nuclear fusion.
Unit-19: Electronic Devices
Semiconductor; semiconductor diode – I – V characteristics in forward and reverse bias, diode as a rectifier; I – V characteristics of LED, photodiode, solar cell, and Zener diode; Zener diode as a voltage regulator. Junction transistor, transistor action, characteristics of a transistor; transistor as an
amplifier (common emitter configuration) and oscillator. Logic gates (OR, AND, NOT, NAND and NOR). Transistor as a switch.
Unit-20: Communication Systems
Elements of a communication system (block diagram only); bandwidth of signals (speech, TV and digital data); bandwidth of transmission medium. Propagation of electromagnetic waves in the atmosphere, sky and space wave propagation. Need for modulation. Production and detection of an amplitude-modulated wave.
AIEEA UG 2015 Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
The Indian Council of Agricultural Research (ICAR) is an autonomous organisation under the Department of Agricultural Research and Education (DARE), Ministry of Agriculture, Government of India.
Syllabus for ICAR AIEEA UG 2015 Entrance Examination are Physics Syllabus, Chemistry Syllabus, Biology Syllabus, Mathematics Syllabus and Agriculture Syllabus.
AIEEA – UG – 2015 Physics Syllabus
AIEEA – UG – 2015 Chemistry Syllabus
AIEEA – UG – 2015 Biology Syllabus
AIEEA – UG – 2014 Agriculture Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1: Agrometeorology, Genetics and Plant Breeding, Biochemistry and Microbiology
Agrometerology: Elements of Weather-rainfall, temperature, humidity, wind velocity, Sunshine weather forecasting, climate change in relation to crop production.
Genetics & Plants Breeding : (a) Cell and its structure, cell division-mitosis and meiosis and their significance (b) Organisation of the genetic materials in chromosomes, DNA and RNA (c) Mendel’s laws of inheritance. Reasons for the success of Mendel in his experiments, Absence of linkage in Mendel’s experiments. (d) Quantitative inheritance, continuous and discontinuous variation in plants. (e) Monogenic and polygenic inheritance. (f) Role of Genetics in Plant breeding, self and cross-pollinated crops, methods of breeding in field crops-introduction, selection, hybridization, mutation and polyploidy, tissue and cell culture, (g) Plant Biotechnology-definition and scope in crop production.
Biochemistry: pH and buffers, Classification and nomenclature of carbohydrates; proteins; lipids; vitamins and enzymes.
Microbiology: Microbial cell structure, Micro-organisms-Algae, Bacteria, Fungi, Actinomycetes, Protozoa and Viruses Role of micro-organisms in respiration, fermentation and organic matter decomposition.
Unit-2: Livestock Production
Scope and importance: (a) Importance of livestock in agriculture and industry. White revolution in India. (b) Important breeds Indian and exotic, distribution of cows, buffaloes and poultry in India.
Care and management: (a) Systems of cattle and poultry housing (b) Principles of feeding, feeding practices. (c) Balanced ration-definition and ingredients. (d) Management of calves, bullocks, pregnant and milch animals as well as chicks crockrels and layers, poultry. (e) Signs of sick animals, symptoms of common diseases in cattle and poultry, Rinderpest, black quarter, foot and mouth, mastitis and haemorrhagic septicaemia coccidiosis, Fowl pox and Ranikhet disease, their prevention and control.
Artificial Insemination: Reproductive organs, collection, dilution and preservation of semen and artificial insemination,
role of artificial insemination in cattle improvement, Livestock Products: Processing and marketing of milk and Milk products.
Unit-3: Crop Production
Introduction: (a) Targets and achievements in foodgrain production in India since independence and its future projections, sustainable crop production, commercialization of agriculture and its scope in India. (b) Classification of field crops based on their utility-cereals, pulses, oils seeds, fibre, sugar and forage crops.
Soil, Soil fertility, Fertilizers and Manures: (a) Soil, soil pH, Soil structure, soil organisms, soil organisms, soil tilth, soil fertility and soil health. (b) Essential plant nutrients, their functions and deficiency symptoms. (c) soil types of India and their characteristics. (d) Organic manure, common fertilizers including straight, complex, fertilizer mixtures and biofertilizers; integrated nutrient management system.
Irrigation and Drainage: (a) Sources of irrigation (rain, canals, rivers, wells, tubewells). (b) Scheduling of irrigation based on critical stages of growth, time interval, soil moisture content and weather parameters. (c) Water requirement of crops. (d) Methods of irrigation and drainage. (e) Watershed management.
Weed Control: Principles of weed control, methods of weed control (cultural, mechanical, chemical, biological and Integrated weed management)
Crops: Seed bed preparation, seed treatment, time and method of sowing/planting, seed rate; dose, method and time of fertilizer application, irrigation, interculture and weed control; common pests and diseases, caused by bacteria, fungi virus and nematode and their control, integrated pest management, harvesting, threshing, post harvest technology: storage, processing and marketing of major field crops-Rice, wheat, maize, sorghum, pearl millet, groundnut, mustard, pigeon-pea, gram, sugarcane, cotton and berseem.
Unit-4: Horticulture
(a) Importance of fruits and vegetables in human diet, Crop diversification & processing Industry. (b) orchard-location and layout, ornamental gardening and kitchen garden. (c) Planting system, training, pruning, intercropping, protection from frost and sunburn. (d) Trees, shrubs, climbers, annuals, perennials-definition and examples. Propagation by seed, cutting, budding, layering and grafting.(e) Cultivation practices, processing and marketing of: (i) Fruits – mango, papaya, banana, guava, citrus, grapes, (ii) Vegetables – Radish, Carrot, potato, onion, cauliflower, brinjal, tomato, spinach and cabbage, (iii) Flowers – Gladiolus, canna, chrysanthemus, roses and marigold. (f) Principles and methods of fruit and vegetable preservation. (g) Preparation of jellies, jams, ketchup, chips and their packing.
AIEEA – UG – 2014 Mathematics Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1: Sets and functions
1. Sets : Sets and their representations. Empty set. Finite & Infinite sets. Equal sets. Subsets, Subsets of the set of real numbers especially intervals (with notations). Power set. Universal set. Venn diagrams. Union and intersection of sets. Difference of sets. Complement of a set.
2. Relations & Functions : Ordered pairs, Cartesian product of sets. Number of elements in the Cartesian product of two finite sets. Cartesian product of the reals with itself (Upto R × R × R). Definition of relation, Types of relations: reflexive, symmetric, transitive and equivalence relations. One to one and onto functions, composite functions, inverse of a function. Binary operations. Pictorial representation of a function, domain. Co-domain and range of a relation. Function as a special kind of relation from one set to another. Real valued function of the real variable, domain and range of these functions, constant, identity, polynomial, rational, modulus, signum and greatest integer functions with their graphs. Sum, difference, product and quotients of functions.
3. Trigonometric Functions: Positive and negative angles. Measuring angles in radians & in degrees and conversion from one measure to another. Definition of trigonometric functions with the help of unit circle. Truth of the identity sin2x + cos2x = 1, for all x. Signs of trigonometric functions and sketch of their graphs. Expressing sin(x +y) and cos (x+y) in terms of sinx, siny, cosx & cosy. Deducing the identities like the following:
Identities related to sin2x, tan2x, sin3x, cos3x and tan3x. General solution of trigonometric equations of the type sin è ? = sin á, cos è ? = cos á? and tan é ? = tan á.
Inverse Trigonometric Functions: Definition, range, domain, principal value branches. Graphs of inverse trigonometric functions. Elementary properties of inverse trigonometric functions.
Properties of triangles, including centroid, incentre, circum-centre and orthocenter, Solution of triangle, Heights and Distances.
Unit-2: Algebra
1. Principle of Mathematical induction: Processes of the proof by induction, motivating the application of the method by looking at natural numbers as the least inductive subset of real numbers. The principle of mathematical induction and simple applications.
2. Complex Numbers and Quadratic Equations: Need for complex numbers, especially-1, to be motivated inability to solve every quadratic equation. Brief description of algebraic properties of complex numbers. Argand plane and polar representation of complex numbers. Statement of Fundamental Theorem of Algebra, solution of quadratic equations in the complex number system.
3. Linear Inequalities: Linear inequalities, Algebraic solutions of linear inequalities in one variable and their representation on the number line. Graphical solution of linear inequalities in two variables. Solution of system of linear inequalities in two variables- graphically.
4. Permutations & Combinations: Fundamental principle of counting. Factorial n.(n!). Permutations and combinations, derivation of formula and their connections, simple applications.
5. Binomial Theorem: History, statement and proof of the binomial theorem for positive integral indices. pascal’s triangle, General and middle term in binomial expansion, simple applications.
6. Sequence and Series: Sequence and Series, Arithematic progression (A.P.). arithmetic mean (A.M.) Geometric progression(G.P), general term of G.P., sum of n terms of a G.P., geometric mean(G.M.). relation between A.M. and G.M. Sum to ne terms of the special series Ón, Ón2, and Ón3.
7. Matrices: Concept, notation, order, equality, types of matrices, zero matrix, transpose of a matrix, symmetric and skew symmetric matrices. Addition, multiplication and scalar multiplication of matrices, simple properties of addition, multiplication and scalar multiplication. None-commutativity of multiplication of matrices and existence of non-zero matrices whose product is the zero matrix(restrict to square matrices of order 2). Concept of elementary row and column operations. Invertible matrices and proof of the unquencess of inverse, if it exists.
8. Determinants: Determinant of a square matrix( up to 3 × 3 matrices), properties of determinants, minors, cofactors and application of determinants in finding the area of a triangle. Adjoint and inverse of a square matrix. Consistency, inconsistency and number of solutions of system of linear equation by examples, solving system of linear equations in two or three variables (having unique solution) using inverse of a matrix.
Unit-3: Coordinate Geometry
1. Straight Lines: Slope of a line and angle between two lines. Various forms of equations of a line: parallel to axes, point-slope form, slope-intercept form, two-point form, intercepts form and normal form. General equation of a line. Distance of a point from a line.
2. Conic Sections: Sections of a cone: circle ellipse, parabola, hyperbola, a point, a straight line and pair of parabola, ellipse and hyperbola. Standard equation of a circle.
3. Introduction to Three-dimensional Geometry: Coordinate axes and coordinate planes in three dimensions. Coordinates of a point. Distance between two points and section formula.
Unit-4: Calculus
1. Limits and Derivatives: Derivative introduced as rate of change both as that of distance function and geometrically, intuitive idea of limit. Definition of derivative, relate it to slope of tangent of the curve, derivative of sum, difference, product and quotient of functions. Derivatives of polynomial and trigonometric functions.
2. Continuity and Differentiability: Continuity and differentiability, derivative of composite functions, chain rule, derivatives of inverse trigonometric functions, derivative of implicit function. Concept of exponential and logarithmic functions and their derivative. Logarithmic differentiation. derivative of functions expressed in parametric forms. Second order derivatives. Rolle’s and Lagrang’s Mean Value Theorems (without proof) and their geometric interpretations.
3. Application of Derivatives: Application of derivatives: rate of change, increasing/decreasing functions, tangents & normals, approximation, maxima and minima(first derivative test motivated geometrically and second derivative test given as a provable tool). Sample problems.
4. Integrals: Integration as inverse process of differentiation. Integration of a variety of functions by substitution, by partial fractions and by parts; only simple integrals of the type
to be a evaluated. Definite integrals as a limit of a sum, Fundamental Theorem of Calculus (without proof). Basic properties of definite integrals and evaluation of definite integrals.
5. Applications of the Integrals : Application in finding the areas under simple curves, especially lines, areas of circles/parabolas/ellipses(in standard form only), area between the two above said curves.
6. Differential Equations: Definition, order and degree, general and particular solutions of a differential equation, Formation of differential equation whose general solution is given. Solution of differential equations by method of separation of variables, homogenous differential equations of first order and first degree. Solutions of linear differential equation of the type: ![]() where p and q are function of x.
where p and q are function of x.
Unit-5: Vectors and Three-Dimensional Geometry
1. Vectors: Vectors and scalars, magnitude and direction of a vector. Direction cosines/ratios of vectors. Types of vectors (equal unit, zero, parallel and collinear vectors), position vector of a pint, negative of a vector, components of a vector, addition of vectors, multiplication of a vector by a scalar position vector of a point dividing a line segment in a given ratio. Scalar (dot) product of vectors, projection of a vector on a line. Vector(cross) product of vectors.
2. Three-dimensional Geometry: Direction cosines/ratios of a line joining two points. Cartesian and vector equation of a line, coplanar and skew lines, shortest distance between two lines. Cartesian and vector equation of a plane. Angle between(i) two lines, (ii) two planes. (iii) a line and a plane. Distance of a point from a plane.
Unit-6: Linear Programming
Linear Programming: Introduction, definition of related terminology such as constraints, objective function, optimization, different types of linear programming (L.P.) problems, mathematical formulation of L.P. problems, graphical method of solution for problems in two variables, feasible and infeasible regions, feasible and infeasible solutions, optimal feasible solutions (up to three non-trivial constraints).
Unit-7: Mathematical Reasoning
Mathematical Reasoning: Mathematically acceptable statements. Connecting words/phrases – consolidating the understanding of “if and only if (necessary and sufficient) condition”, “implies”, “and/or”, “implied by”, “and” “or”, “there exists” and their use through variety of examples related to real life and Mathematics Validating the statements involving the connecting words, difference between contradiction, converse and contra positive.
Unit-8: Statistics & Probability
1. Statistics: Measures of central tendency, mean, median and mode from ungrouped/grouped data. Measures of dispersion, mean deviation, variance and standard deviation from ungrouped/ data. Correlation, regression lines.
2. Probability: Random experiments: outcomes, sample spaces (set representation) Events: occurrence of events, ‘not’, ‘and’ and ‘or’ events, exhaustive events, mutually exclusive events Axiomatic (set theoretic) probability, Probability of an event, probability of ‘not’, ‘and’ & ‘or’ events. Multiplication theorem on probability. Conditional probability, independent events, total probability. Bayes’ theorem, Random variable and its probability distribution, mean and variance of stochastic variable. Repeated independent (Bernoulli) trials and Binomial distribution.
Unit-9: Statics
Introduction: basic concepts and basic laws of mechanics, force resultant of forces acting at a point, parallelogram law of forces, resolved parts of a force, Equilibrium of a particle under three concurrent forces. Triangle law of forces and its converse, Lami’s theorem and its converse, Two parallel forces, like and unlike parallel forces, couple and its moment.
Unit-10: Dynamics
Speed and velocity, average speed, instantaneous speed, acceleration and retardation, resultant of two velocities. Motion of a particle along a line, moving with constant acceleration. Motion under gravity. Laws of motion, Projectile motion.
AIEEA – UG – 2014 Biology Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit: 1 The living World
Nature and scope of Biology, Methods of Biology. Our place in universe. Laws that govern the universe and life. Level of organization. Cause and effect relationship.
Origin of life and its maintenance. Origin and diversity of life. Physical and chemical principles that maintain life processes. The living crust and interdependence. The positive and negative aspects of progress in biological sciences. The future of the living world. identification of human responsibility in shaping our future.
Unit:2 Unit of Life
Cells as a unit of life. Small biomolecules; water, minerals, mono and oligosaccharides, lipids, amino acids, nucleotides and their chemistry, cellular location and function. Macromolecules in cells – their chemistry, cellular location and functional significance, Polysaccharides, proteins and nucleic acids. Enzymes; chemical nature, classification, mechanism in action-enzyme complex, allosteric modulation (brief), inrreversible activation. Biomembranes; Fluid mosaic model of membrane, role in transport, recognition of external information (brief). Structural organization of the cell; light and electron microscopic view of cell, its organelles and their functions; nucleus mitochondria, chloroplasts, endoplasmic reticulum. Golgi complex, lysosomes, microtubules, cell wall, cilia and flagella, vacuoles, cell inclusions. A general account of cellular respiration. Fermentation, biological oxidation (A cycle outline), mitochondrial electron transports chain, high energy bonds and oxidative phosphorylation, cell reproduction; process of mitosis and meiosis.
Unit-3: Diversity of Life
Introduction. The enormous variety of living things, the need for classification to cope with this variety; taxonomy and phylogeny; shortcomings of a two kingdom classification as plants and animals; the five kingdom classification, Monera, Protista, Plantae, Fungi and Animalia; the basic features of five kingdom classification. Modes of obtaining nutrition-autotrophs and heterotrophs. Life style producers, consumers and decomposers. Unicellularity and multicellularity, phyylogenetic relationships. Concepts of species, taxon and categories – hierarchical levels of classification; binomial nomenclature; principles of classification and nomenclature; identification and nature of viruses and bacteriophages; kingdom Monera-archeabacteria – life in extermen environments; Bacteria, Atinomycetes, Cyanobacteria. Examples & illustration of autotrophic and heterotrophic life; mineralizes-nitrogen fixers; Monera in cycling matter; symbiotic forms; disease producers. Kingdom Protissta-Eukaryotic unicellular organisms, development of flagella and cilia; beginning of mitosis; syngamy and sex. Various life styles shown in the major phyla. Evolutionary precursors of complex life forms. Diatoms, dinoflagellates, slime moulds, protozons; symbiotic forms. Plant kingdom-complex autotrophs, red brown and green algae; conquest of land, bryophytes, ferns, gymnosperms and angiosperms. Vascularization; development of flower, fruit and seed. Kingdom fungi-lower fungi(Zygomycetes), higher fungi (Ascomycetes and Basidiomycetes); the importance o fungi. Decomposers; parasitic forms; lichens and mycorrhizae. Animal kingdom-animal body pattern and symmetry. The development of body cavity in invertebrate vertebrate physia. Salient features with reference to habitat and example of phylum porifera, coelenterate, helminthes, annelids, mollusca, arthropoda, echinoderms; chordate (classes-fishes, amphibians, reptiles, birds and mammals) highlighting major characters.
Unit:4 Organisms and Environment
Species : Origin and concept of species population, interaction between environment and population community. Biotic community, interaction between different species, biotic stability. Changes in the community. Succession, Ecosystem; interaction between biotic and abiotic components; major ecosystems, manmade ecosystem – Agro ecosystem. Biosphere; flow of energy, trapping of solar energy, energy pathway, food web, biogeochemial cycles, calcium and sulphur, ecological imbalance and its consequences. Conservation of natural resources’ nerewable and non-renewable (in brief). Water and land management, wasteland development. Wild life and forest conservation; causes for the extinction of some wild life, steps taken to conserve the remaining species, concept of endangered concept of afforestation. Environmental pollution; air and water pollution, sources, major pollutants of big cities of our country, their effects and methods of control, pollution due to fallout and waste disposal, effect and control, noise pollution; sources and effects.
Unit:5 Multicellulairty : Structure and Function – Plant Life
Form and function. Tissue system in flowering plants; meristematic and permanent. Mineral nutrition-essential elements, major functions of different elements, passive and active uptake of minerals. Modes of nutrition, transport of solutes and water in plants. Photosynthesis; photochemical and biosynthetic phases, diversity in photosynthetic pathways, photosynthetic electron transport and photophosphorylation, photorespiration. Transpiration and exchange of gases. Stomatal mechanism. Osmoregulation in plants: water relations in plant cells, water potential. Reproduction and development in Angiosperms; asexual and sexual reproduction. Structure and functions of flower : development of male and female gametophytes in angiosperms, pollination, fertilization and development of endosperm, embryo seed and fruit. Differentiation and organ formation. Plant hormones and growth regulation; action of plant hormones in relation to seed dormancy and germination, apical dominance. senescence and abscission. Application of synthetic growth regulators. A brief account of growth and movement in plants.
Unit:6 Multicellularity : Structure and Function – Animal Life
Animal tissues, epithelial, connective, muscular, nerve. Animal nutrition, organs of digestion and digestive process, nutritional requirements for carbohydrates, proteins, fats, mineral and vitamins; nutritional imbalances and deficiency diseases. Gas exchange and transport: Pulmonary gas exchange and organs involved, transport of gases in blood, gas exchange in aqueous medic circulation: closed and open vascular systems, structure and pumping action of heart, arterial blood pressure, lymph. Excretion and osomoregulation. Ammonotelism, Ureotelism, urecotelism, excretion of water and urea with special reference to man. Role of kidney in regulation of plasma, osmolarity on the basis of nephron structure, skin and lungs in excretion. Hormonal coordination; hormones of mammals, role of hormones as messengers and regulators. Nervous coordination, central autonomic and peripheral nervous systems, receptors, effectors, reflex action, basic physiology of special senses, integrative control by neuroendocrinal systems. Locomotion: joints, muscle movements, types of skeletal muscles according to types of movement, basic aspects of human skeleton. Reproduction; human reproduction, female reproductive cycle. Embryonic development in mammals (upto three germs layers), growth, repair and ageing.
Unit:7 Continuity of Life
Heredity and variation: Introduction, Mendel’s experiments with peas and concepts of factors. Mendel’s laws of inheritance. Genes: Packaging of heredity material in prokaryotes-bacterial chromosome and plasmid; and eukaryote chromosomes. Extranuclear genes, viral genes. Linkage (genetic) maps. Sex determination and sex linkage. Genetic material and its replication, gene manipulation, Gene expression; genetic code, transcription, translation, gene regulation. Molecular basis of differentiation.
Unit:8 Origin and Evolution of Life
Origin of life: living and non-living, chemical evolution. organic evolution; Oparin ideas, Miller-Urey experiments. Interrelationship among living organisms and evidences of evolution: fossil records including geological scale, Morphological evidence – hematology, vestigial organs, embryological similarities and biogeographical evidence.
Darwin’s two major contributoions. Common origin of living organisms and recombination as sourece of variability selection and variation, adaptation (Lederberg’s replica plating experiment for indirect selection of bacterial mutants), reproductive isolation, speciation. Role of selection, change and drift in determining composition of population. Selected examples: industrial melanism; drug resistance, mimicry, malaria in relation to G-6-PD deficiency and sickle cell disease. Human evolution: Placontological evidence, man’s place among mammals. Brief idea of dryopithecus, Australopithecus, Homo erectus. H. neanderthlensis, Cro-Magnon man and Homo sapiens. Human chromosomes, similarity in different racial groups. Comparison with chromosomes of non-human primates to indicate common origin; Cultural vs. biological evolution.
Mutation: origin and types of mutation, their role in speciation.
Unit:9 Application of Biology
Introduction, role of biology, in the amelioration of human problems. Domestication of plant- a historical account, improvement of crop plants; Principles of plant breeding and plant introduction. Use of fertilizers, their economic and ecological aspects.
Use of pesticides: advantages and hazards. Biological methods of pest control. Crops today. Current concerns, gene pools and genetic conservation. Underultilized crops with potential uses of oilseeds, medicines, beverages, spices, fodder. New crops-Leucaena (Subabul), Jojoba, Guayule, winged bean, etc. Biofertilizers – green manure, crop residues and nitrogen fixation (symbiotic, non symbiotic). Applications of tissue culture and genetic engineering in crops. Domestication and introduction of animals. Livestock, poultry, fisheries (Fresh water, marine, aquaculture). Improvement of animals: principles of animal breeding. Major animal diseases and their control. Insects and their products (silk, honey, wax and lac). Bioenergy-biomass, wood(combustion; gasification, ethanol). Cow dung cakes, gobar gas, plants as sources of hydrocarbons, for producing petroleum, ethanol from starch and lignocelluloses. Biotechnology, application in health and agriculture, genetically modified (GM) organisms, bio-safety issues. A brief historical account-manufacture of cheese. yoghurt, alcohol, yeast, vitamins, organic acids, antibiotics, steroids, dextrins. Scalling up laboratory findings to industrial production, sewage treatment. Production in insulin, human growth hormones, interferon. Communicable diseases including STD and diseases spread through blood transfusion (hepatitis, AIDS, etc) Immune response, vaccine and antisera. Allergies and Inflammation. Inherited diseases and dysfunction, sex-linked diseases, genetic incompatibilities, and genetic counseling. Cancer-major types. causes, diagnosis and treatment. Tissue and organ transplantation. Community health services and measures; blood banks; mental health, smoking, alcoholism and drug addiction-physiological symptoms and control measures. Industrial wastes, toxicology, pollution-related diseases. Biomedical engineering – spare parts for man, instruments for diagnosis of diseases and care. Human population related diseases. Human population, growth, problems and control, inequality between sexes, control measures; test-tube babies aminocentesis. Future of Biology.
AIEEA – UG – 2014 Chemistry Syllabus
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
Unit-1 : Some Basic Concepts of Chemistry
General Introduction: Importance and scope of chemistry. Historical approach to particulate nature of matter, laws of chemical combination. Dalton’s atomic theory: concept of elements, atoms and molecules. Atomic and molecular masses mole concept and molar mass: percentage composition, empirical and molecular formula chemical reactions, stoichiometry and calculations based on stoichiometry.
Unit-2: Solid State
Classification of solid based on different binding forces : molecular, ionic, covalent and metallic solids, amorphous and crystalline solids (elementary idea), unit cell in two dimensional and three dimensional lattices, calculation of density of unit cell, packing in solids, voids, number of atoms per unit cell in a cubic unit cell, point defects, electrical and magnetic properties.
Unit-3: Solutions
Types of solutions, expression of concentration of solutions of solids in liquids, solubility of gases in liquids, solid solutions, colligative properties-relative lowering of vapour pressure, elevation of Boiling Point, depression of freezing point, osmotic pressure, determination of molecular masses using colligative properties, abnormal molecular mass.
Unit-4: Structure of Atom
Discovery of electron, proton and neutron; atomic number, isotopes and isobars. Thomson’s model and its limitations, Rutherford’s model and its limitations. Bohr’s model and its limitations, concept of shells and subshells, dual nature of matter and light, de Broglie’s relationship. Heisenberg uncertainty principle, concept of orbitals, quantum numbers, shapes of s, p, and d orbitals, rules for filling electrons in orbitals-Aufabau principle, Pauli exclusion principle and Hund’s rule, electronic configuration of atoms, stability of half filled and completely filled orbitals.
Unit-5: Classification of Elements and Periodicity in Properties
Significance of classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements –atomic radii, ionic radii. Ionization enthalpy, electron gain enthalpy, electro negativity, valence.
Unit-6: Chemical Bonding and Molecular Structure
Valence electrons, ionic bond, covalent bond: bond parameters. Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR(Valence shell electron pair repulsion) theory, concept of hybridization, involving s, p and d orbitals and shapes of some simple molecules, molecular orbitas; theory of homonuclear diatomic molecules (qualitative idea only), hydrogen bond.
Unit-7: States of Matter: Gases and Liquids
Three states of matte. Intermolecular interactions, type of bonding, melting and boiling points. Role of gas laws in elucidating the concept of the molecule, Boyle’s law. Charles law, Gay Lussac’s law, Avogadro’s law. Ideal behavior, empirical derivation of gas equation, Avogadro’s number. Ideal gas equation. Derivation from ideal behavior, liquefaction of gases, critical temperature. Liquid State- Vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations)
Unit-8: Thermodynamics
Concepts of System, types of systems, surroundings, Work, heat, energy, extensive and intensive properties, state functions. First law of thermodynamics – internal energy and enthalpy, heat capacity and specific heat, measurement of DU and DH, Hess’s law of constant heat summation, enthalpy of: bond dissociation, combustion, formation, atomization, sublimation. Phase transformation, ionization, and solution. Introduction of entropy as a state function, free energy change for spontaneous and non-spontaneous processes, criteria for equilibrium.
Unit-9: Equilibrium
Equilibrium in physical and chemical processes, dynamic nature of equilibrium, law of mass of action, equilibrium constant, factors affecting equilibrium – Le Chatelier’s principle; ionic equilibrium – ionization of acids and bases, strong and weak electrolytes, degree of ionization, concept of pH. Hydrolysis of salts. Buffer solutions, solubility product, common ion effect.
Unit-10: Redox Reactions
Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, applications of redox reactions.
Unit-11: Hydrogen
Position of hydrogen in periodic table, occurrence, isotopes, preparation, properties and uses of hydrogen; hydrides – ionic, covalent and interstitial; physical and chemical properties of water, heavy water; hydrogen peroxide-preparation, properties and structure; hydrogen as a fuel.
Unit-12: s-Block Elements (Alkali and Alkaline earth metals)
Group 1 and Group 2 elements
General introduction, electronic configuration, occurrence, anomalous properties of the first element each group, diagonal relationship, trends in the variation of properties (such as ionization enthalpy, atomic and ionic radii), trends in chemical reactivity with oxygen, water, hydrogen and halogens; uses.
Unit-13: Preparation and properties of some important compounds
Sodium carbonate, sodium chloride, sodium hydroxide and sodium hydrogen carbonate, biological importance of sodium and potassium. CaO, CaCO3 and industrial use of lime and limestone, biological importance of Mg and Ca
Unit-14: Some p-Block Elements
General Introduction to p-Block Elements: Group 13 elements
General introduction, electronic configuration, occurrence. Variation of properties, oxidations states, trends in chemical reactivity, anomalous properties of first element of the group; Boron-physical and chemical properties, some important compound: borax, boric acids, boron hydrides. Aluminum: uses, reactions with acids and alkalies.
Unit-15: Group 14 elements
General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous behavior of first element, Carbon – catenation, allotropic forms, physical and chemical properties; uses of some important compounds: oxides. Important compounds of silicon and a few uses: silicon tetrachloride, silicones, silicates and zeolites.
Unit-16: Organic Chemistry
Some Basic Principles and Techniques
General Introduction, methods of qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds. Electronic displacements in a covalent bond: inductive effect, electromeric effect, resonance and hyper conjugation. Homolytic and heterolytic fission of a covalent bond: free radicals, carbocations, carbanions; electrophiles and nucleophiles, types of organic reactions.
Unit-17: Hydrocarbons
Classification of hydrocarbons
Alkanes – Nomenclature, isomerism, conformations (ethane only), physical properties, chemical reactions including free radical mechanism of halogenation, combustion and pyrolysis.
Alkenes – Nomenclature, structure of double bond (ethane) geometrical isomerism, physical properties, methods of preparation; chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikov’s addition and peroxide effect), ozonolysis, oxidation, mechanism of electrophilic addition.
Alkynes – Nomenclature, structure of triple bind (ethyne), physical properties. Methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of – hydrogen, halogens, hydrogen halides and water.
Aromatic hydrocarbons: Introduction, IUPAC nomenclature; benzene: resonance, aromaticity, chemical properties mechanism of electrophilic substitution. – nitration, sulphonation, halogenations. Friedel-Craft’s alkuylation and acylation: directive influence of functional group in mono-substituted benzene; carcinogenicity and toxicity.
Unit-18: Electrochemistry
Conductance in electrolytic solutions, specific and molar conductivity variations of conductivity with concentration, Kohlrausch’s Law, electrolysis and laws of electrolysis (elementary idea), dry cell – electrolytic cells and Galvanic cells: lead accumulator, EMF of a cell, standard electrode potential, Nernst equation and its application to chemical cells, fuel cells; corrosion.
Unit-19: Chemical Kinetics
Rate of a reaction (average and instantaneous), factors affecting rate of reaction; concentration, temperature, catalyst; order and molecularity of a reaction; rate law and specific rate constant, integrated rate equations and half life (only for zero and first order reactions); concept of collision theory (elementary idea, no mathematical treatment)
Unit-20: Surface Chemistry
Adsoprtion – physisorption and chemisorptions; factors affecting adsorption of gases on solids; catalysis : homogenous and heterogeneous, activity and selectivity: enzyme catalysis; colloidal state: distinction between true solutions, colloids and suspensions; lyophilic lyophobic, multimolecular and macromolecular colloids; properties of colloids; Tyndall effect, Brownian movement, electrophoresis, coagulation; emulsion – types of emulsions.
Unit-21: General Principles and Processes of Isolation of Elements
Principles and methods of extraction – concentration, oxidation, reduction electrolytic method and refining; occurrence and principles of extraction of aluminium, copper, zinc and iron.
Unit-22: p-Block Elements
Group 15 elements
General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; nitrogen – preparation, properties and uses; compounds of nitrogen: preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous-allotropic froms; compounds of phosphorous: preparation and properties of phosphine, halides(PCl3, PCl5) and oxoacids
Unit-23: Group 16 elements
General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; simple oxides; Ozone. Sulphur-allotropic forms; compounds of sulphur: preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Unit-24: Group 17 elements
General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds, oxoacids of halogens (structures only).
Unit-25: Group 18 elements
General introduction, electronic configuration. Occurrence, trends in physical and chemical properties, uses.
Unit-26: d and f Block Elements
General introduction, electronic configuration, occurrence and characteristics of transition metals. General trends in properties of the first row transition metals – metallic character, ionization enthalpy, oxidation states, ionic radii, colour catalytic property, magnetic properties, interstitial compounds, alloy formation preparation and properties of K2Cr2O7 and KMnO4.
Lanthnoids – electronic configuration, oxidation states, chemical reactivity and lanthanoid contraction.
Actinoids – Electronic configuration, oxidation states.
Unit-27: Coordination Compounds
Coordination compounds – Introduction, ligands, coordination number, colour, magnetic properties and shapes, IUPAC nomenclature of mononuclear coordination compounds, bonding; isomerism, importance of coordination compounds (in qualitative analysis, extraction of metals and biological systems).
Unit-28: Haloalkanes and Haloarenes
Halokanes : Nomenclature, nature of C-X bond, physical and chemical properties, mechanism of substitution reactions.
Halorenes : Nature of C-X bond, substitution reactions (directive influence of halogen for monosubstituted compounds only) Uses and environmental effects of – dichloromethane, trichloromethane, tetrachloromethane, iodoform, freons, DDT.
Unit-29: Alcohols, Phenols and Ethers
Alcohols
Nomenclature, methods of preparation, physical and chemical properties (of primary alcohols only); identification of primary, secondary and tertiary alcohols; mechanism of dehydration, uses of methanol and ethanol. Phenols : Nomenclature, methods of preparation, physical and chemical properties, acidic nature of phenol, electrophillic substitution reactions, uses of phenols, Ethers: Nomenclature, methods of preparation, physical and chemical properties, uses,
Unit-30: Aldehydes, Ketones and Carboxylic Acids
Aldehydes and Ketones: Nomenclature, nature of carbonyl group, methods of preparation, physical and chemical properties mechanism of nucleophillic addition, reactivity of alpha hydrogen in aldehydes; uses.
Carboxylic Acids: Nomenclature, acidic nature, methods of preparation, physical and chemical properties; uses.
Unit-31: Organic compounds containing Nitrogen
Amines: Nomenclature, classification, structure, methods of preparation, physical and chemical properties, uses, identification of primary, secondary and tertiary amines.
Cyanides and Isocyanides – will be mentioned at relevant places in context.
Diazonium salts: Preparation, chemical reactions and importance in synthetic organic chemistry.
Unit-32: Biomolecules
Carbohydrates – Classification (aldoses and ketoses), monosaccharide (glucose and fructose), oligosaccharides (sucrose, lactose, maltose), polysaccharides (starch, cellulose, glycogen); importance
Proteins – Elementary idea of a-amino acids, peptide bond, polypeptides, proteins, structure of amines-primary, secondary, tertiary structure and quaternary structures (qualitative idea only), denaturation of proteins; enzymes.
Vitamins – Classification and functions.
Nucleic Acids: DNA and RNA.
Unit-33: Polymers
Classification- natural and synthetic, methods of polymerization (addition and condensation), copolymerization. Some important polymers: natural and synthetic like polythene, nylon, polyesters, Bakelite, rubber.
Unit-34: Environmental Chemistry
Environmental pollution – air,, water and soil pollution, chemical reactions in atmosphere, smog, major atmospheric pollutants; acids rain, ozone and its reactions, effects of depletion of ozone layer, greenhouse effect and global warming – pollution due to industrial wastes; green chemistry as an alternative tool for reducing pollution, strategy for control of environmental pollution.
Unit-35: Chemistry in Everyday life
1. Chemical in Medicines – analgesics, tranquilizers, antiseptics, disinfectants, antimicrobials, antifertility drugs, antibiotics, antacids, antihistamines.
2. Chemical in food – preservatives, artificial sweetening agents.
3. Cleansing agents – soaps and detergents, cleansing action.
AIEEA – UG – 2014 Exam Pattern
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
- The examination shall be conducted in one of the two subject streams namely
‘Stream-A’ comprising of Physics, Chemistry and Agriculture/Biology subjects.
‘Stream-B’ comprising of Physics,Chemistry and Mathematics subjects.
- Questions for Physics and Chemistry subjects shall be common for both subject streams.
- Paper for ‘Stream–A’ shall carry questions on Biology as well as Agriculture subject, out of which the candidate has to attempt/opt/choose only one subject, either Biology or Agriculture. ‘Stream–B’ paper carries questions on Mathematics subject.
- AIEEA Question Paper will have 180 Multiple Choice type questions, each with four options.
- 60 questions in Physics, serially numbered from 1-60,
- 60 questions in Chemistry, numbering from 61-120
- 60 questions in Agriculture/Biology/Mathematics, numbering from 121-180.
- Each correctly answered question will earn four marks, with a maximum of 720 marks for the paper.
- The duration of the exam would be 2½ hours.
- The Entrance Examination
- For each incorrect answer one mark will be deducted from the total score.
- Question with no response indicated will not be awarded any mark and there will be no negative marking for that question.
- The candidates are advised not to attempt such questions in the OMR answer sheet, for which they are not sure of the correct answer.
- More than one answer indicated against a question will be deemed as incorrect answer and will invite negative marking.
- Candidate will be required to choose the correct answer and mark in the OMR answer sheet by darkening the corresponding circle/ bubble against the serial number of the question with black/ blue ball-point pen.
-
Question paper for the examination shall be bilingual, i.e., in ENGLISH and HINDI. In case of any variation in English and Hindi versions of question(s), English version will be taken as the final and correct version.
-
A visually challenged (blind) candidate, can request for the services of amanuensis (scribe) who must be one grade junior in academic qualification i.e. not more than X/ Matriculation pass in the present case and should not be related to the candidate. For amanuensis, the candidate must submit a separate request letter, at least one week in advance, to the concerned Nodal officer of the Examination City Centre giving a copy of the PD/PC certificate issued by a competent authority. The services of amanuensis (Scribe) who should be from academic discipline other than that of the candidate, shall be provided free of cost by the Nodal Officer.
- The scribe will have to give a suitable undertaking, confirming that the scribe fulfills all the stipulated eligibility criteria for a scribe as mentioned above. In case later on it is proved that candidate has suppressed material facts, the candidature of the applicant will stand cancelled, irrespective of the outcome of result of the written test. Such candidates who use a scribe shall be eligible for extra time of 50 minutes during examination.
AIEEA – UG – 2014 Conduct of Examination
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
(i) Examination Hall will be opened to candidates 30 minutes before the commencement of examination.
(ii) Candidate who does not possess Admit Card (candidate copy), duly attested and signed will not be admitted to the Examination Hall under any circumstances.
(iii) Pre-written material in any form, viz., notes/ books/ log table, and mobile phones/i-pods, calculator or any electronic device are not allowed inside the examination hall. For verification, candidate may be searched at any time during the examination.
(iv) The examination will start strictly on time and an announcement to this effect will be made by the Invigilator.
(v) A time signal will be given ringing a long bell at the start of the examination. To indicate that one hour is remaining, or 15 minutes are remaining, short bells will be given. A signal (long bell) will also be given at the closing time for collection of OMR sheet and question booklet when the candidate must stop writing/marking the
answers.
(vi) Candidate coming late may be allowed to enter the examination hall up to 10.30 A.M. only but no extra time will be given. Candidate coming late beyond 30 minutes, after the commencement of the examination, will not be permitted.
(vii) Fifteen minutes before the commencement of the examination, each candidate will be given OMR Answer sheet and five minutes before, the Question Booklet.
(viii) Immediately on receipt of Question Booklet, candidate should write/fill-in the required particulars on the cover page of the Question Booklet with BLACK/ BLUE BALL-POINT PEN ONLY. Candidate will not open the Question Booklet, until asked to do so by the Invigilator.
(ix) Candidate will write required particulars on OMR answer sheet with only BLACK / BLUE BALLPOINT PEN on SIDE-I and –II.
(x) No candidate shall be allowed to leave the Examination Hall until the completion of examination and related formalities. Candidate will hand over Question Booklet and OMR Answer Sheet to the Invigilator before leaving the Examination Hall.
(xi) After completing the examination and before handing over the Question Booklet and the OMR Answer Sheet, the candidate should once again check that all the particulars required in the Question Booklet and the OMR Answer Sheet has been correctly filled-in.
(xii) The OMR Answer sheet shall be scanned and evaluated through computer and the category- wise final merit list for counselling shall be prepared based on the reservation category information given by the candidate in OMR Answer sheet. It must be ensured by the candidate that Roll Number, Subject Stream, Series of Question Booklet, optional paper attempted and Reservation Category are correctly written in the OMR Answer Sheet and also the OMR Answer Sheet is duly signed by
the invigilator and the candidate at appropriate places, failing which it may not be considered for evaluation.
(xiii) Before leaving the Examination Hall, the candidate must ensure that OMR Answer Sheet and Question Booklet have been handed over to the invigilator on duty. In case, the candidate does not hand over the OMR Answer Sheet with Question Booklet to the invigilator and takes away the same with him/her, this shall amount
to use of unfair means and the candidate will be declared failed besides inviting further necessary action.
(xiv) Candidates are advised to bring with them their own writing material such as hard card-board (on which nothing should be written) and their own good quality black/blue ball-point pens.
(xv) Any attempt of using unfair means by the candidate during the examination process will render him/her liable to be disqualified and his/her her candidature for the examination would be forfeited.
AIEEA – UG – 2014 Online Application
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
- This is an alternative option to OMR forms. Online Application and downloadable prospectus will be available on ICAR website. A Demo Online application form will also be available on the website for acquainting the candidates with this technique and for practicing before online submission of application.
- The candidate has to visit www.icar.org.in and click the option of filling online application AIEEA- UG-2014. The candidate can download prospectus and then fill his/her from online. He/ she need not purchase the separate or fill the form manually on OMR Sheet.
- During filling of prescribed online form, candidate can either opt for making fee payment through Bank
- Challan of Syndicate Bank or through Credit / Debit card using Payment Gateway of Syndicate Bank/NEFT.
- Making payment through Bank Challan
After filling the online application, candidate has to take a printout of computer generated confirmation page and bank challan (in 3 copies) through which he / she has to deposit cash (fees) in the nearest Syndicate Bank branch. Bank will return two copies of challan duly stamped. Candidate will retain one copy of the challan. He / she will enclose the second copy (duly stamped by the bank ) with his her filled in computer generated confirmation page’s printout and send it to COE (Edn.) by post at the given address.
- Making payment through Credit/Debit card using Payment Gateway of Syndicate Bank During online filling of the form, candidate can also opt for making fee payment through Credit / Debit card using Payment Gateway of Syndicate Bank. In this case, the candidate will make payment directly through the debit/ credit card from his computer and will not be required to visit the bank.
- Receipt of such payments/Application is confirmed immediately.
- Making payment through NEFT While filling the form the candidates may also opt for ‘Payment through NEFT’. When candidates have selected this option, after filling the form, a NEFT Challan shall be generated. The candidates can take a print out of this Challan and send NEFT remittance through any branch of any bank. In such cases, the candidates must fill the information as available in the NEFT Challan generated through ICAR website. The candidates must obtain the UTR number from the Bank through which they have sent NEFT remittance.
- The candidates are required to enclose a photocopy of the Banks’ challan bearing the UTR number along with the application without fail.
- After obtaining the UTR number, the candidates are required to visit the ICAR website again and upload the UTR number details in the website. Candidates are requested to ensure that only correct UTR details are entered in the website as the in-correct details shall lead to rejection of the application without further correspondence in the matter. No Bank charges are payable for NEFT payment.
- While filling the Online form,Candidate can upload soft copy of his /her clear, recent, colour photograph, same as pasted on Application Form, signature and thumb impression. If he / she is unable to do so then he / she has to paste the photograph, put his / her signature and thumb impression on the printout of computer generated confirmation page in designated boxes and send it to COE (Edn.) by post at the given address.
- Application will be allowed either on OMR application form attached with the Information Bulletin AIEEAUG 2014 or ONLINE only. No application will be allowed on any other format and will be summarily rejected.
AIEEA – UG – 2014 Evaluation and Declaration of Result
Click here for Stream A Model Papers
Click here for Stream A Preparatory Course
Click here for Stream B Model Papers
Click here for Stream B Preparatory Course
(i) OMR answer sheets of the candidates shall be scanned through computer/scanner and evaluated through computer by matching the OMR answer sheet responses of the candidate with the Stream-wise (Stream-A/ Stream-B), optional subject-wise (Agriculture/ Biology/ Mathematics) and Series-wise (Series-1/ Series-2) Answer Key templates prepared beforehand. Overall merit-rank list shall be prepared separately for both subject streams. In the event of candidates getting equal marks in the Entrance Examination, relative merit will be determined on the basis of marks obtained in the compulsory subjects (Physics and Chemistry) in AIEEA-UG Examination.
In the event of tie again, the ratio of positive and negative marks will be decided. Candidate with higher absolute value of the ratio will be given better rank. In the event of tie again, a candidate, higher in age, would be rated higher in merit. Category-wise final merit list for counseling shall be prepared based on the reservation category information given by the candidate in OMR Answer sheet.
(ii) The result of the AIEEA-UG-2014 is likely to be declared after third week of May 2014 and will be placed on ICAR website. The copies of the result will also be displayed at the Examination Cell of the Indian Council of Agricultural Research, Krishi Anusandhan Bhavan-II, Pusa, New Delhi-110 012.
(iii) The result of the entrance examination would be available in terms of overall merit-rank obtained by the candidates qualified for counselling.
(iv) All announcements related to the conduct of entrance examination including, issue of examination notification,admit card information examination result, call letter for counseling, general notices, etc. would be posted regularly and available on the ICAR website. The candidates are advised to be vigilant about the announcements on the ICAR website as the ICAR would not be held responsible for non-receipt of any information/ letter by post or otherwise.
(v) No separate intimation about the non-selection of the candidate in AIEEA-UG-2014 Examination, nor marks obtained therein, will be sent to the candidate and no correspondence (Letter/ Fax/ e-mail/ Telegram) in this regard will be entertained.